Your shopping cart is currently empty
Your shopping cart is currently empty
HSP90 Is Essential for Hepatitis A Virus Replication
Understanding Host Factors in Viral Replication
Viruses rely heavily on host cellular machinery to complete their life cycles. Among the most important host factors are molecular chaperones—proteins that assist in the folding, stabilization, and assembly of other proteins. Increasing evidence suggests that many viruses exploit these cellular systems to ensure the proper formation of viral proteins and replication complexes.
A recent study published in the Journal of Virology (J Virol. 2025 Jun 5;99(7):e00502-25. doi: 10.1128/jvi.00502-25 ) highlights a previously underappreciated role for the molecular chaperone HSP90 in the replication of Hepatitis A virus (HAV). Although HAV is a well-known cause of acute viral hepatitis worldwide, the host factors required for efficient viral replication remain incompletely understood.
HSP90 Plays a Critical Role in HAV Replication
The researchers found that HSP90 activity is essential for efficient HAV replication. When the function of this molecular chaperone is disrupted, viral replication levels drop significantly in cellular models.
HSP90 is a central regulator of protein homeostasis within the cell. As a molecular chaperone, it assists client proteins in achieving and maintaining their functional conformations. Many signaling proteins, including kinases and transcription factors, rely on HSP90 for stability.
The study suggests that HAV exploits this host chaperone system to support its own replication process. By utilizing the cellular protein-folding machinery, the virus can stabilize viral proteins and promote the formation of replication complexes necessary for viral genome amplification.
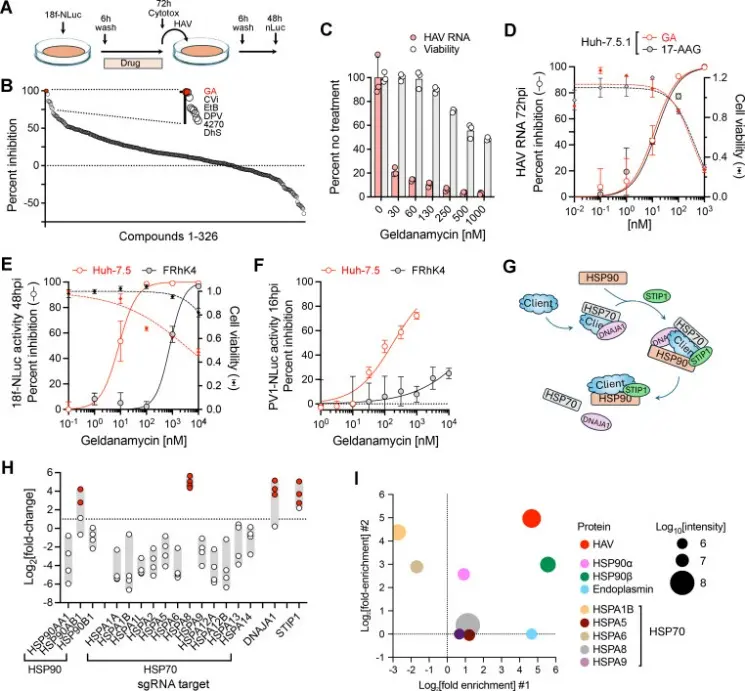
Fig.1 The heat shock pathway is required for HAV replication.
Chaperone Systems as Emerging Targets in Antiviral Research
These findings highlight a broader principle in virology: host protein-folding networks are often critical enablers of viral replication. Molecular chaperones such as HSP90 have been implicated in the life cycles of multiple RNA viruses.
As researchers continue to explore virus–host interactions, host chaperone systems are emerging as important nodes that viruses exploit during infection. Understanding these dependencies may help identify new strategies for studying viral replication and host-directed antiviral approaches.
Studies like this deepen our understanding of the complex relationship between viral pathogens and the cellular systems they rely on.
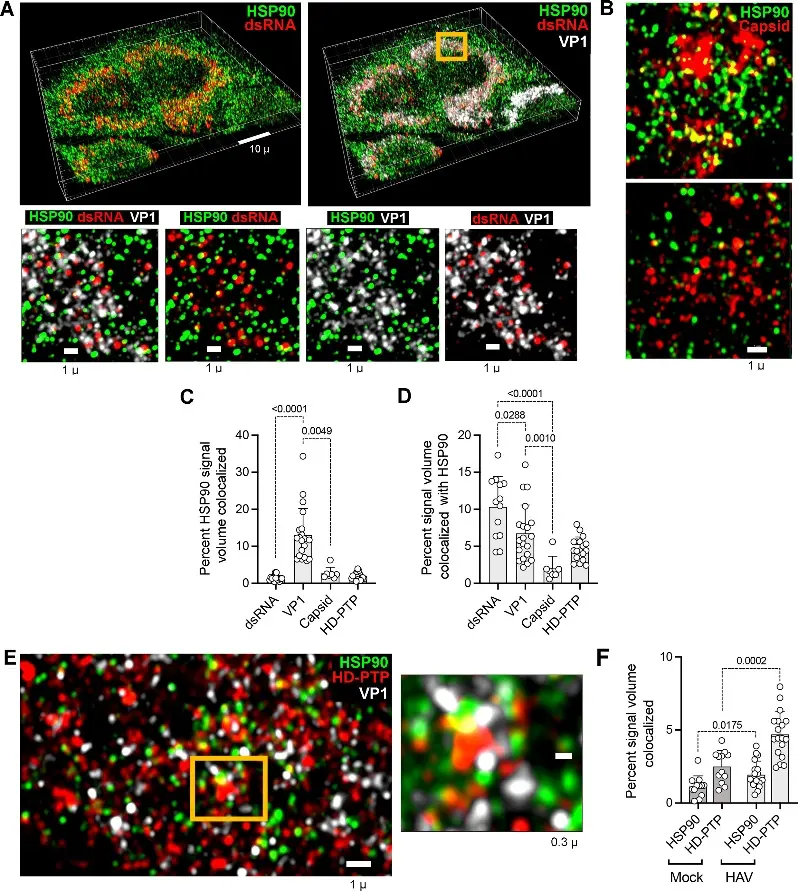
Fig.2 Airyscan super-resolution confocal immunofluorescence microscopy of Huh-7.5 cells infected with HM175/p16 virus for 7 days.
Source:
Li Y, Zheng X, Xie L, Kapustina M, Shirasaki T, Yonish B, Chen X, Hirai-Yuki A, Nagata N, Suzuki R, Isogawa M, Vogt MR, Muramatsu M, Lemon SM.2025.Heat shock protein 90 chaperone activity is required for hepatitis A virus replication. J Virol99:e00502-25.https://doi.org/10.1128/jvi.00502-25
Researchers interested in studying host–virus interactions and antiviral mechanisms can explore additional research tools and resources related to HSP90 and viral replication on our website.
Other Articles

Subscription to TargetMol News
An essential round-up of science news, opinion and analysis, delivered to your inbox every weekday.

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.