Your shopping cart is currently empty
Your shopping cart is currently empty

Molecular Binding & Interaction Assay
SPR, BLI & biophysical assays integrated.Binding dynamics decoded.


Molecular Binding & Interaction Assay
For a candidate drug, target binding is a prerequisite for its biological function.
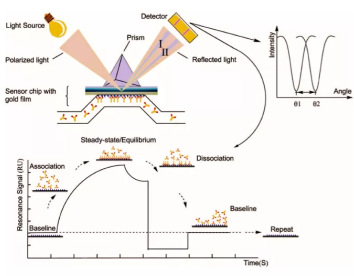
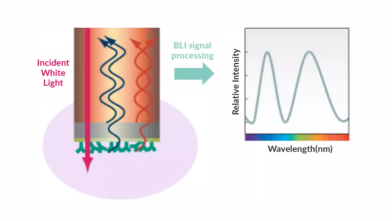
Biacore assays, developed based on SPR (Surface Plasmon Resonance) technology, enable highly sensitive, real-time monitoring of binding and interactions between two molecules, and are compatible with high-throughput screening. As an advanced biosensor-based analytical technique, SPR is applied throughout every stage of drug discovery and development, including target identification, compound screening, proteomics, immunogenicity assessment, biologic drug development and manufacturing, as well as broader life science research.
TargetMol offers a wide range of in vitro services for evaluating binding interactions between small-molecule drugs and target proteins, including:
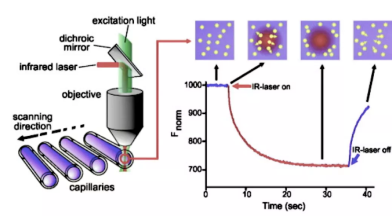
SPR (Surface Plasmon Resonance)/MST (Microscale Thermophoresis)/BLI (Bio-Layer Interferometry)/ITC (Isothermal Titration Calorimetry)/DSF (Differential Scanning Fluorimetry)

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.