Your shopping cart is currently empty
Your shopping cart is currently empty
Breakthrough in Cell: Decoding the Century-Old Mystery of Nicotine Biosynthesis
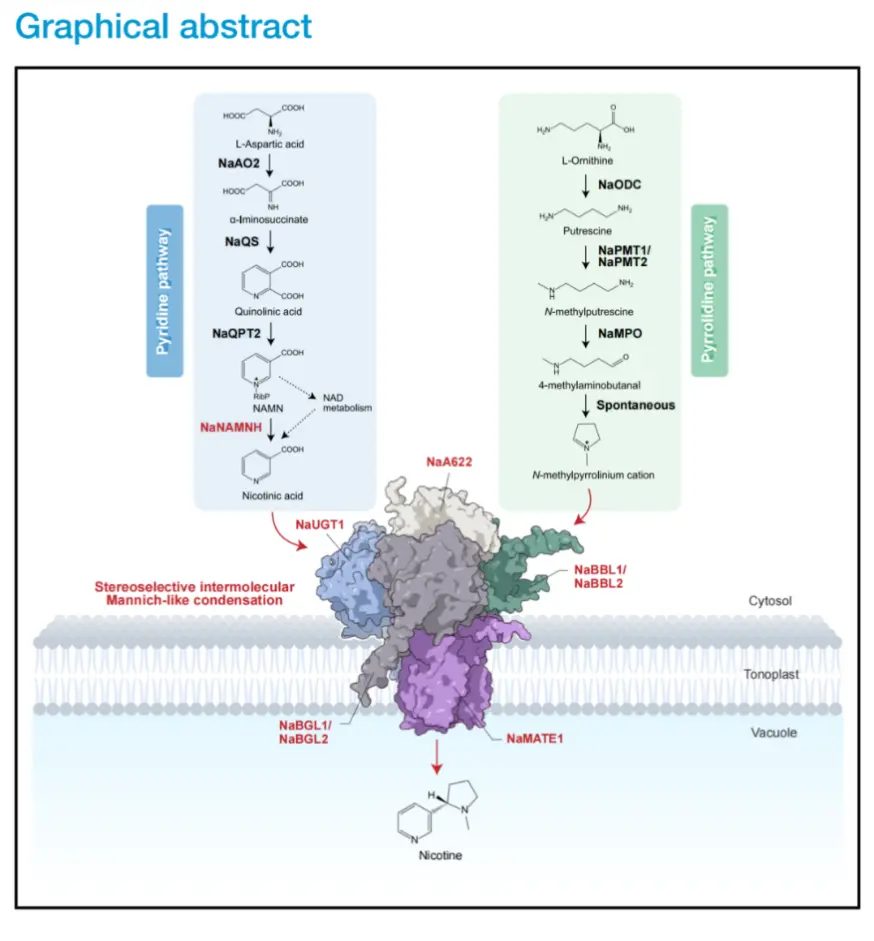
Overview
A groundbreaking study titled "Complete biosynthesis of nicotine", published in the prestigious journal Cell, has finally mapped the full biosynthetic pathway of nicotine. This research, led by the CAS Centre for Excellence in Molecular Plant Sciences, deciphers the chemical coupling of nicotine’s two heterocyclic rings and reveals how plant cells achieve flawless coordination between substrate transport and biochemical reactions through a sophisticated “vacuolar membrane metabolon”. This “glycosylation-guided, deglycosylation-driven” paradigm fundamentally reshapes our understanding of metabolic spatial-temporal regulation, providing a new theoretical framework for the evolution and synthesis of defensive secondary metabolites in plants.
The "Glycosylation Switch" Paradigm
The study shifts the traditional focus from free nicotinic acid (NA) to its glucose-conjugated form. Using information-theory-guided omics, the researchers identified NaUGT1, a glycosyltransferase that converts NA into nicotinic acid glucose ester (NA-Glc). This glycosylation is not a detoxification pathway but a strategic metabolic "activation" that prepares the substrate for the final assembly.
The Vacuolar Membrane Metabolon
The most significant discovery is the identification of a five-component metabolon anchored to the vacuolar membrane (tonoplast). This molecular machinery consists of:
• Transporters: NaMATE1 and NaMATE2, which facilitate the movement of intermediates.
• Enzymatic Core: The β-glucosidases NaBGL1/2 (responsible for deglycosylation) and the berberine bridge-like enzymes NaBBL1/2/3 (responsible for condensation).
This spatial organization ensures substrate channeling, allowing the plant to achieve high-efficiency, stereoselective synthesis while sequestering reactive intermediates to avoid cellular toxicity.
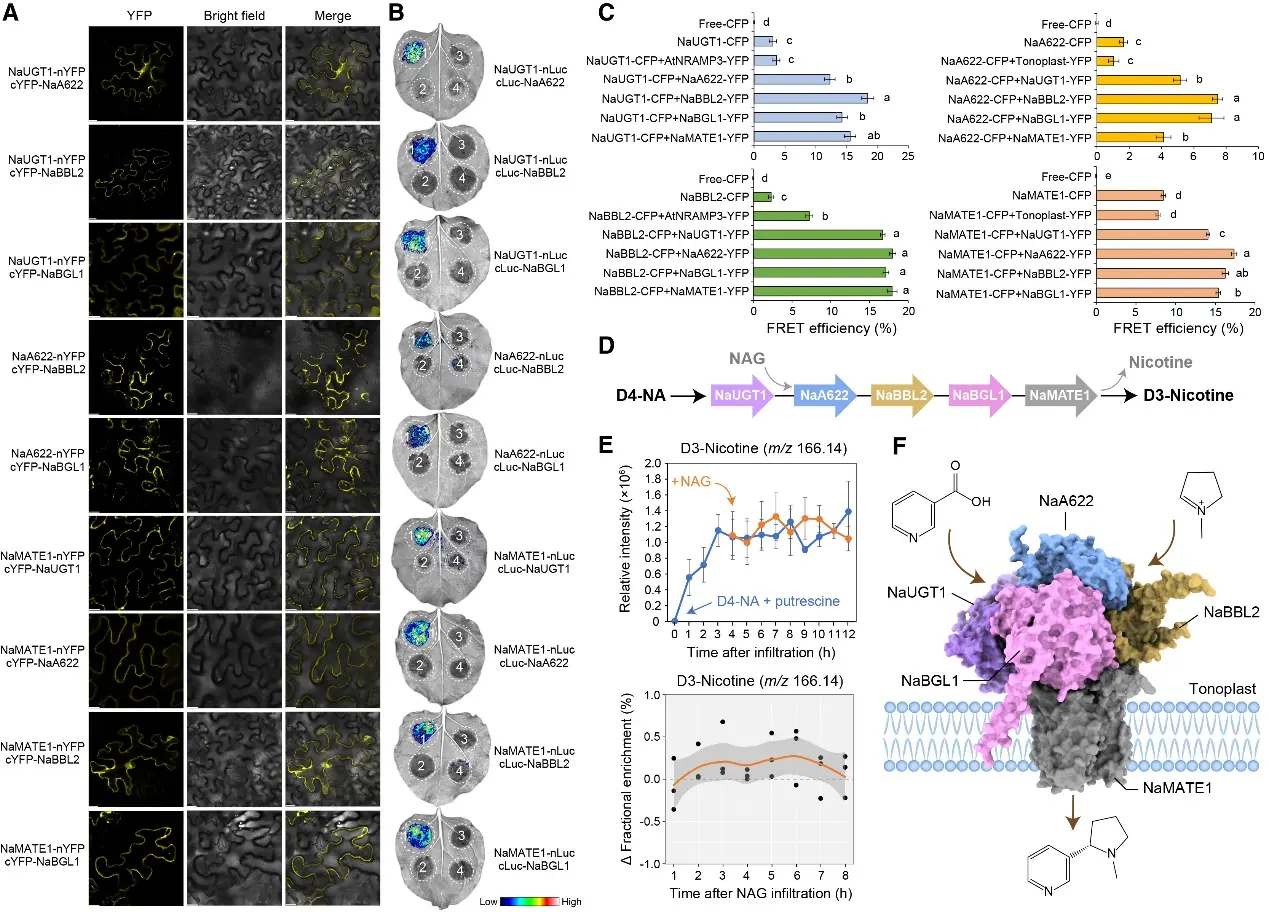
Figure 1 Protein-protein interactions in planta reveal that multi-enzyme protein complexes form at vacuolar membranes, facilitating channeled nicotine biosynthesis and transport
Metabolic Flux Validation
A critical phase of the research involved confirming that NA-Glc is indeed the physiological precursor for nicotine in vivo. To achieve this, the team performed isotope-labeled metabolic flux analysis. By supplying tobacco tissues with D4-nicotinic acid, they tracked the isotopic signature as it moved from NA to NA-Glc and was ultimately incorporated into the nicotine molecule. The precise detection of these D4-labeled intermediates provided the empirical evidence needed to validate the glycosylation-deglycosylation model.
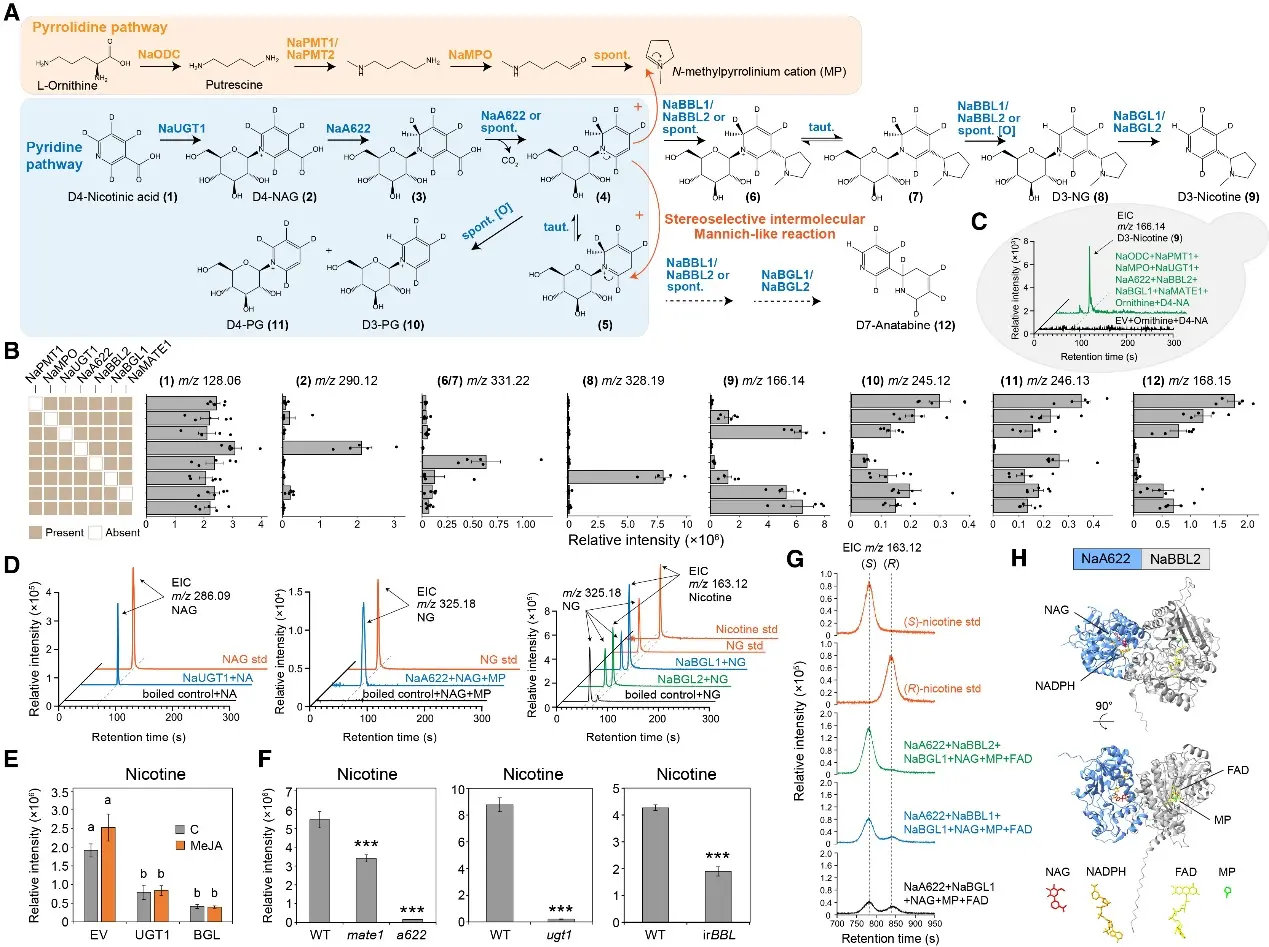
Figure 2 Biosynthesis and engineering of nicotine in vitro and in vivo
*All images are sourced from the original publication.
Broader Implications
The study successfully reconstructed this complex pathway in heterologous systems, such as tomato and microbes, demonstrating the portability of the tonoplast-localized metabolon. This provides a programmable blueprint for the synthetic biology of alkaloids and enhances our understanding of how plants evolve complex defense mechanisms through spatial and temporal metabolic regulation.
Other Articles

Subscription to TargetMol News
An essential round-up of science news, opinion and analysis, delivered to your inbox every weekday.

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.