Your shopping cart is currently empty
Your shopping cart is currently empty
TargetMol—Disease Modeling—PMA (Cat. No. TQ0198, Cas. 16561-29-8), Classical PKC Activator
1. Product Introduction
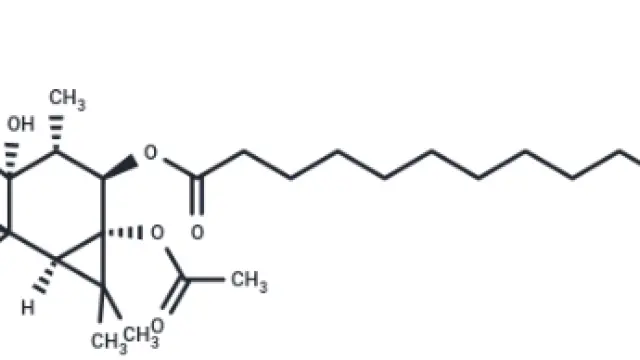
Phorbol 12-myristate 13-acetate (Cat. No. TQ0198, Cas. 16561-29-8), also known as PMA. PMA is a phorbol ester natural product, which can be used as an activator of PKC, SphK and NF-κB. It is often used to induce THP-1 cell differentiation and construct dermatitis models.
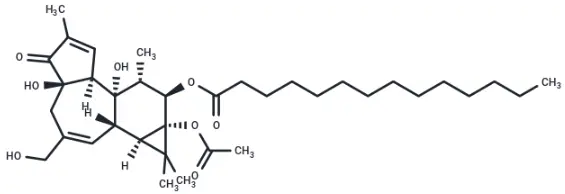
Molecular structure of PMA
2. Background Introduction
PMA is a typical phorbol ester natural product, which can directly activate the PKC signaling pathway as a diacylglycerol (DAG) analogue, triggering a series of intracellular cascade reactions. Studies have shown that PMA can strongly promote the translocation and activation of various subtypes of PKC (especially PKCα, PKCδ, etc.) to the cell membrane, and then regulate the expression of downstream transcription factors and inflammation-related genes, induce cell cycle arrest, oxidative stress response and cytoskeleton remodeling. In the THP-1 monocyte model, PMA promotes the transformation of cells from a suspended growth state to an adherent macrophage-like phenotype by activating PKC and related signal networks, accompanied by up-regulation of differentiation markers such as CD11b and CD14 and enhanced phagocytosis. Therefore, it is widely used to construct a monocyte-macrophage differentiation model and inflammation-related research system in vitro [1]. In addition, PMA can further amplify the inflammatory response by inducing ROS production and regulating pathways such as NF-κB, thus playing a role as a ' inflammatory stimulator ' in a variety of immune and inflammatory models. [2]
3. Application References
Phorbol myristate acetate induces differentiation of THP-1 cells in a nitric oxide-dependent manner

Research Overview:
In this study, the mechanism of phorbol 12-myristate 13-acetate (PMA) inducing THP-1 monocytes to differentiate into macrophage-like cells was investigated, and the regulatory role of nitric oxide (NO) in this process was studied. By stimulating THP-1 cells with PMA, it was observed that the cell adhesion was enhanced, the morphology gradually changed from a suspended round to a adherent, spread-out macrophage-like phenotype, accompanied by changes in differentiation-related functions. At the mechanism level, the experimental results showed that the PMA-induced differentiation process was accompanied by a significant increase in the level of NO production, and inhibition of nitric oxide synthase (NOS) activity could significantly weaken the PMA-induced cell differentiation effect, indicating that NO signal plays a key regulatory role in this process. In addition, studies have also shown that calcium signaling may be involved in the synergistic regulation of NO production and differentiation. It is speculated that calcium signaling may enhance the iNOS/NO pathway and mediate PMA-induced THP-1 cell differentiation. [1]
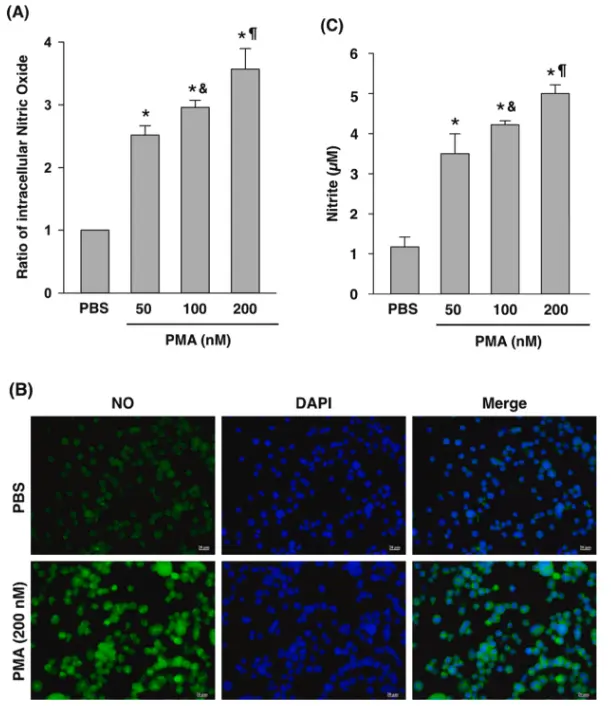
PMA increases nitric oxide ( NO ) concentration [1]
Resting time after phorbol 12-myristate 13-acetate in THP-1 derived macrophages provides a non-biased model for the study of NLRP3 inflammasome

Research Overview:
This study mainly explored the effect of 'resting time' on the reliability of the NLRP3 inflammasome research model during PMA-induced THP-1 monocyte differentiation into macrophage-like cells. Firstly, THP-1 cells were treated with PMA to differentiate into macrophage-like state, and the basic inflammation level of cells and the response to NLRP3 inflammasome stimulation under different recovery time conditions were further systematically compared. The results showed that THP-1 cells were induced to differentiate with 5 ng / mL PMA, and the 24 h resting phase provided a model similar to macrophages at the morphological and molecular levels. However, even at low concentrations, PMA can induce IL-1β production. The 24 h rest period down-regulated the expression of pro-IL-1β in the PMA-rest group, but did not affect its ability to respond to the second stimulus by activating the inflammasome. [3]
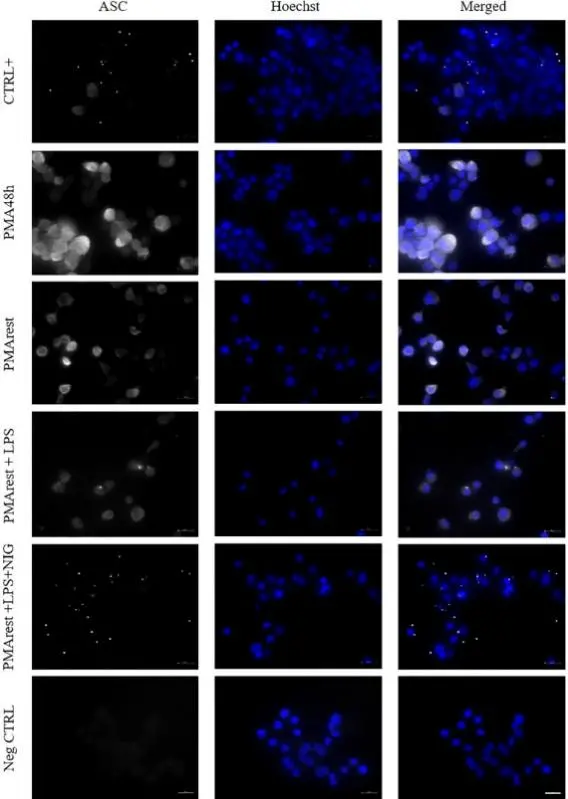
Immunofluorescence analysis of ASC spots [3]
Elevated miR-17-5p facilitates mycobacterial immune evasion by targeting MAP3K2 in macrophages

This study found that elevated miR-17-5p in macrophages weakens the host 's innate immune defense against mycobacterial infection by targeting and inhibiting MAP3K2 expression, thereby promoting the immune escape of pathogens. The authors demonstrated that up-regulation of miR-17-5p inhibits MAP3K2-mediated downstream signal transduction, reduces the production of inflammatory factors and antibacterial effects, and weakens the ability of macrophages to clear mycobacteria. Interfering with miR-17-5p or restoring MAP3K2 expression can enhance the antibacterial response and limit bacterial survival. This study reveals the molecular mechanism of miRNA regulating host signaling pathways involved in Mycobacterium immune escape, and provides a new potential strategy for enhancing anti-infective immunity by targeting the miR-17-5p / MAP3K2 axis. [4]
In this study, Phorbol 12−myristate 13−acetate (PMA) was used as a macrophage activation and differentiation stimulant. In this study, we established a stable and controllable macrophage function model by PMA treatment to induce cell differentiation into macrophage-like phenotype and activate related signaling pathways, which was used to evaluate the regulatory effect of miR-17-5p on MAP3K2 signaling and anti-mycobacterial immune response. PMA is used as an experimental immune stimulation tool to enhance and standardize the response state of macrophages.
4. References
[1] Chang YY, Lu CW, Jean WH, Shieh JS, Lin TY. Phorbol myristate acetate induces differentiation of THP-1 cells in a nitric oxide-dependent manner. Nitric Oxide. 2021 May 1;109-110:33-41. doi: 10.1016/j.niox.2021.02.002. Epub 2021 Mar 2. PMID: 33667621.
[2] Traore K, Trush MA, George M Jr, Spannhake EW, Anderson W, Asseffa A. Signal transduction of phorbol 12-myristate 13-acetate (PMA)-induced growth inhibition of human monocytic leukemia THP-1 cells is reactive oxygen dependent. Leuk Res. 2005 Aug;29(8):863-79. doi: 10.1016/j.leukres.2004.12.011. Epub 2005 Feb 24. PMID: 15978937.
[3] Giambelluca S, Ochs M, Lopez-Rodriguez E. Resting time after phorbol 12-myristate 13-acetate in THP-1 derived macrophages provides a non-biased model for the study of NLRP3 inflammasome. Front Immunol. 2022 Dec 22;13:958098. doi: 10.3389/fimmu.2022.958098. PMID: 36618426; PMCID: PMC9817155.
[4] Jahan MS, Yang J, Tang Y, Xiong D, Hu P, Tang X, Tang L. Elevated miR-17-5p facilitates mycobacterial immune evasion by targeting MAP3K2 in macrophages. Front Immunol. 2025 Dec 4;16:1676204. doi: 10.3389/fimmu.2025.1676204. PMID: 41425567; PMCID: PMC12711757.
Other Articles
Subscription to TargetMol News
An essential round-up of science news, opinion and analysis, delivered to your inbox every weekday.

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.