Your shopping cart is currently empty
Your shopping cart is currently empty
Caspases in Cell Death and Inflammation:
Mechanisms, Pathways, and Research Tools

Caspases are a family of cysteine-dependent proteases that play central roles in programmed cell death and inflammatory signaling. Initially recognized as the key executioners of apoptosis, Caspases are now known to regulate multiple forms of cell death, including pyroptosis, while also participating in immune and inflammatory responses.
Due to their broad biological significance, Caspases have become important research targets in oncology, neurodegenerative diseases, autoimmune disorders, and inflammation-related studies.
Core Mechanism of Caspases
Caspases are highly conserved proteases that specifically cleave substrates following aspartic acid residues. Under physiological conditions, they exist as inactive precursors known as procaspases. Upon cellular stress or death signaling, procaspases undergo proteolytic activation and initiate cascade amplification events that ultimately determine cell fate.
Depending on the signaling context and Caspase subtype involved, activation may result in apoptotic cell death, inflammatory cytokine maturation, or pyroptotic membrane rupture and immune activation.
Functional Classification of Caspases
Although Caspases share similar enzymatic activity, they perform distinct biological functions.
Initiator Caspases
Initiator Caspases include Caspase-2, -8, -9, and -10. These proteins contain extended prodomains such as DED or CARD motifs that enable them to sense upstream death signals, including death receptor activation and mitochondrial cytochrome c release.
Once activated, initiator Caspases trigger downstream signaling cascades by cleaving effector Caspases.
Effector Caspases
Effector Caspases, including Caspase-3, -6, and -7, are responsible for executing apoptosis. Activated effector Caspases cleave structural and regulatory proteins such as PARP and nuclear lamins, leading to characteristic apoptotic morphology and irreversible cellular dismantling.
Inflammatory Caspases
Inflammatory Caspases include Caspase-1, -4, -5, and -11. These enzymes are closely associated with inflammasome activation and inflammatory signaling.
Their major functions include:
• processing IL-1β and IL-18 into mature cytokines
• cleaving Gasdermin D
• inducing pyroptosis through membrane pore formation
Because of their involvement in innate immunity and inflammatory disease, inflammatory Caspases are widely studied therapeutic targets.
Apoptosis vs. Pyroptosis: Why Caspases Matter
Different Caspase pathways can lead to fundamentally different biological outcomes.
Apoptosis is generally considered a controlled and non-inflammatory form of programmed cell death that maintains tissue homeostasis. In contrast, pyroptosis is a highly inflammatory process characterized by membrane rupture and cytokine release.
Understanding which Caspase pathway is activated is therefore critical for interpreting disease mechanisms and designing therapeutic strategies.
Strategies for Targeting Caspases
Selecting an appropriate Caspase-targeting strategy depends on the experimental objective and pathway of interest.
Pan-Caspase Inhibitors
Pan-Caspase inhibitors broadly suppress Caspase activation across multiple pathways.
Main advantages
• strong inhibition of the entire Caspase cascade
• effective reversal of Caspase-dependent cell death phenotypes
• robust cytoprotective effects
Applications
• ischemia and neuroprotection studies
• validation of Caspase-dependent phenotypes
• investigation of alternative cell death pathways
Selective Caspase Inhibitors or Activators
Selective modulators target individual Caspase subtypes and provide greater mechanistic resolution.
Main advantages
• high target specificity
• improved pathway discrimination
• precise interrogation of apoptosis or pyroptosis signaling
Applications
• inflammasome and pyroptosis research
• pathway-specific mechanistic studies
• targeted anti-cancer apoptosis research
Common Methods for Measuring Caspase Activity
Caspase activity is commonly assessed using substrate cleavage assays and protein-based detection methods.
Fluorometric or Luminescent Assays
These assays use labeled peptide substrates such as DEVD-based substrates for Caspase-3/7 detection. Cleavage of the substrate generates measurable fluorescent or luminescent signals that reflect enzymatic activity.
Western Blot Analysis
Western blotting remains a standard method for validating Caspase activation through detection of:
• cleaved Caspase fragments
• procaspase depletion
• downstream substrate cleavage such as PARP
Combining functional and protein-level assays often provides more reliable pathway validation.
TargetMol Caspase Research Tools
TargetMol provides a range of Caspase-targeting compounds and apoptosis-related research tools for pathway investigation and drug discovery applications.
Commonly used products include:
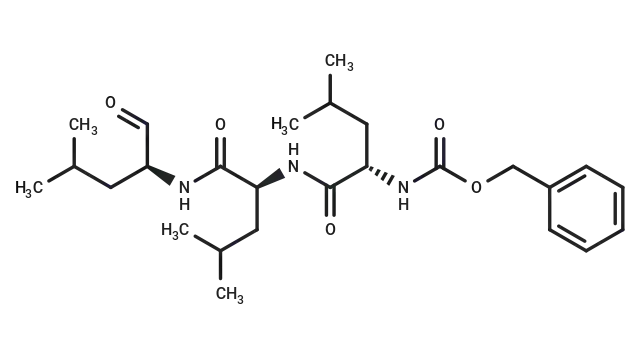
• [T7020] Z-VAD(OH)-FMK – An irreversible pan-Caspase inhibitor that can be directly used in culture media without esterase pre-treatment
• [T6013] Z-VAD(OMe)-FMK – A cell-permeable irreversible pan-Caspase inhibitor widely regarded as a gold-standard tool for apoptosis validation
• [T6005] Z-DEVD-FMK – A cell-permeable and highly selective Caspase-3 inhibitor
• [T7019] Z-IETD-FMK – A cell-permeable and highly selective Caspase-8 inhibitor that blocks the extrinsic apoptosis pathway
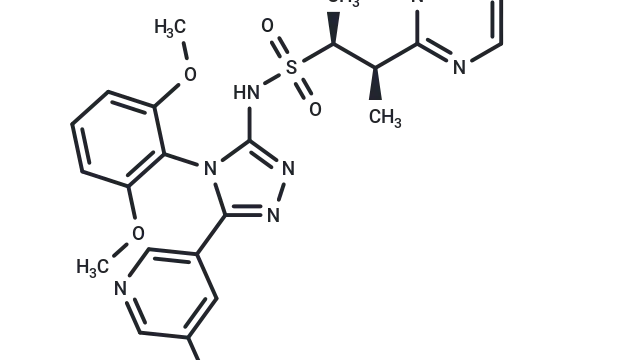
• [T6090] Belnacasan (VX-765) – A potent and selective Caspase-1/4 inhibitor commonly used to inhibit pyroptosis and IL-1β release
• [L9000] Apoptosis Compound Library – A curated collection of apoptosis-related compounds containing numerous active small molecules targeting Caspases and apoptosis pathways
Interested in these Bcl-2 family products?
Visit our website for full product details or get in touch with our team at sales@targetmol.com .
Other Articles
Subscription to TargetMol News
An essential round-up of science news, opinion and analysis, delivered to your inbox every weekday.

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.