Your shopping cart is currently empty
Your shopping cart is currently empty
TargetMol—Alkaloid—Cycloheximide (Cat. No. T1225, CAS. 66-81-9), a Natural Product with Antifungal Activity
1. Product Introduction
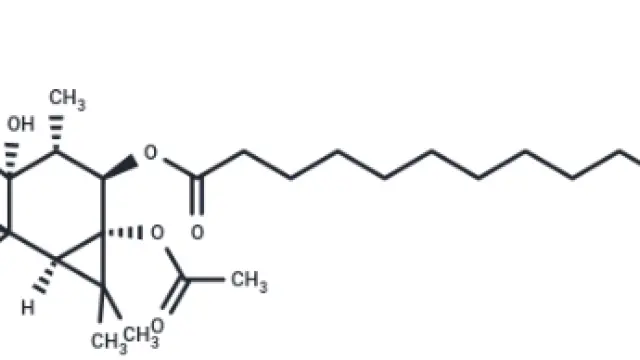
Cycloheximide (Cat. No. T1225, CAS. 66-81-9), also known as Naramycin A, CHX, Actidione. Cycloheximide (Naramycin A) is a natural product with IC50 values of 532.5 nM and 2880 nM for in vivo protein synthesis and RNA synthesis, respectively. Cycloheximide is an antifungal antibiotic that inhibits ferroptosis and inhibits autophagy.
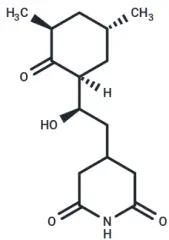
Molecular structure of Cycloheximide
2. Background Introduction
Cycloheximide (CHX) is a natural antifungal antibiotic produced by the soil actinomycete Streptomyces griseus, which is widely used in biomedical research as a protein synthesis inhibitor in eukaryotic cells. It can effectively block the elongation stage in the eukaryotic translation process, thereby inhibiting the overall protein synthesis. This feature makes it an important experimental tool for determining protein half-life, translation regulation and related cellular mechanism research.
The molecular mechanism of cycloheximide is mainly to block the translation elongation stage of eukaryotic ribosomes. It prevents translation elongation factor-mediated mRNA / tRNA translocation by binding to a specific site on the ribosomal large subunit (60S), stopping the ongoing peptide chain elongation, thereby inhibiting new protein synthesis. Classical studies have shown that the position of cycloheximide is closely related to translocation during elongation and can stabilize the polyribosome state. It is a commonly used drug tool for intracellular translation analysis and half-life determination. Because it inhibits overall protein synthesis, it can also indirectly inhibit autophagy by increasing intracellular free amino acid levels and activating the mTORC1 signaling pathway under specific stress conditions. For example, in the starvation-induced autophagy model, Cycloheximide inhibits the formation of autophagy markers by activating mTORC1. [1]
3. Application References
Pharmacokinetics of cycloheximide in rats and evaluation of its effect as a blocker of intestinal lymph formation
Research Overview:
This study systematically evaluated the pharmacokinetic characteristics of Cycloheximide in rats and verified its effectiveness as an ' intestinal lymphatic formation blocker ' in vivo experiments. The key pharmacokinetic parameters such as plasma concentration-time curve, clearance rate, distribution volume and bioavailability of CHX were determined by different routes of administration, and the characteristics of rapid distribution and clearance in vivo were clarified. On this basis, the study further used a lymphatic catheter intubation model to experimentally evaluate the inhibitory effect of CHX on intestinal chylomicron formation and lymphatic lipid transport. The results showed that CHX could significantly inhibit the protein synthesis of intestinal epithelial cells, thereby effectively blocking the formation of chylomicrons and significantly reducing the amount of lipids and drugs transported through the lymphatic pathway. [2]
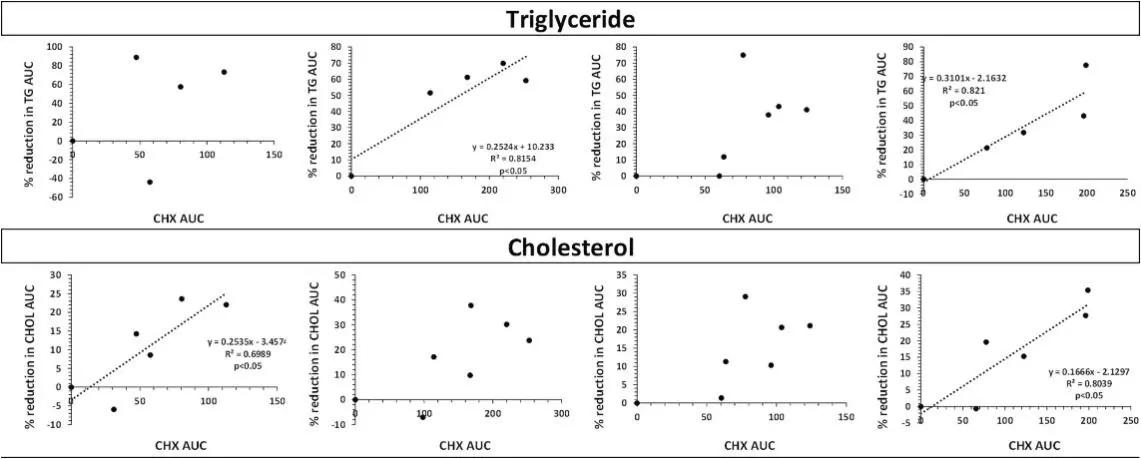
Lipid concentration after oral administration of peanut oil, oral administration of CHX 2.5 mg/kg and no oral administration of CHX 2.5 mg/kg. [2]
Cycloheximide-resistant ribosomes reveal adaptive translation dynamics in C. elegans

Research Overview:
By constructing a cycloheximide-insensitive ribosome mutant model in C.elegans, this study bypassed the systematic bias of traditional CHX treatment on translation kinetics research. After obtaining CHX-resistant ribosomes by genetic engineering, combined with ribosome sequencing and transcriptome analysis, the changes of translation elongation rate, ribosome stagnation site and gene-specific translation efficiency were compared with or without CHX intervention. The results showed that conventional CHX treatment could reshape the distribution pattern of ribosomes on mRNA, thus concealing the real dynamic process of translation. In the context of CHX-resistant, it can reveal the characteristics of translation extension closer to reality in physiological state. This study not only proves the experimental artifacts that may be introduced by CHX in translation studies, but also provides a new strategy to analyze the mechanism of translation regulation in vivo by using drug-resistant ribosome models, which is of great methodological significance for ribosome sequencing methodology and translation kinetics research. [3]
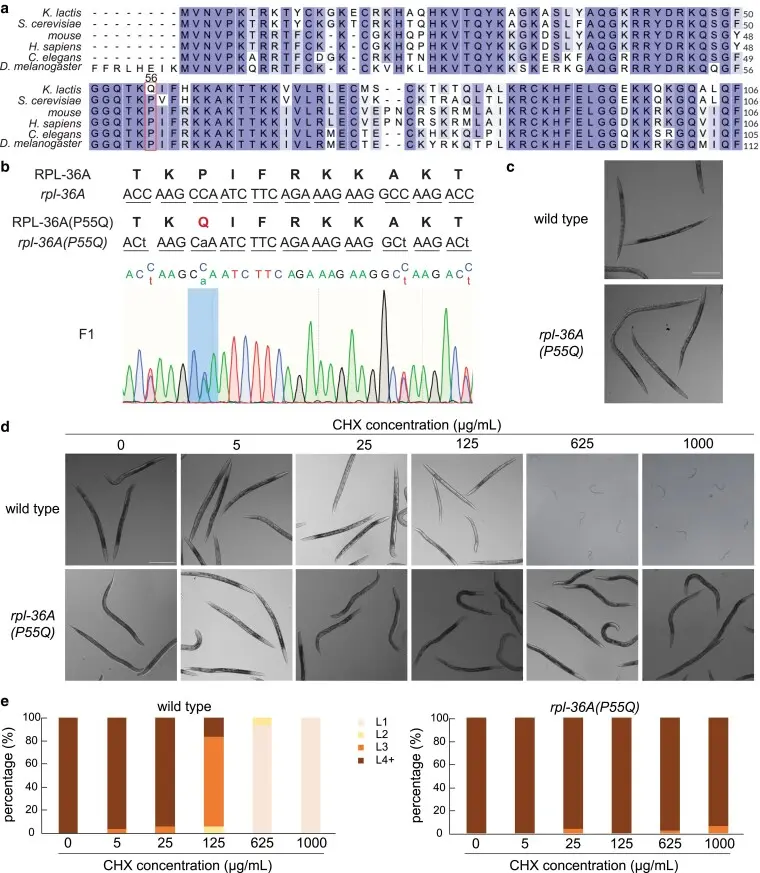
The Rpl-36A ( P55Q ) mutant was resistant to high concentration of CHX. [3]
Salvianolic acid C protects against sepsis-associated acute kidney injury through promotion of PGC1α-mediated renal gluconeogenesis

This study focused on the key pathological link of mitochondrial metabolic disorder in sepsis-associated acute kidney injury ( SA-AKI ), and found that impaired renal gluconeogenesis is an important metabolic basis for sepsis-induced renal failure. The authors confirmed that Salvianolic acid C can significantly improve the energy metabolism of renal tubular epithelial cells under sepsis conditions through in vivo and in vitro models. The core mechanism is to up-regulate the expression and activity of the main metabolic regulator PGC1α, restore the inhibited renal gluconeogenesis pathway, and then improve mitochondrial function and reduce oxidative stress and cell damage. Further molecular experiments showed that the recovery of PGC1α was the key link in the renal protective effect of Salvianolic acid C, and the protective effect was significantly weakened after inhibiting PGC1α. [4]
In this study, Cycloheximide was used as a protein synthesis inhibitor. Its main purpose was to block the translation of nascent proteins to determine whether Salvianolic acid C up-regulates PGC1α and downstream gluconeogenesis-related proteins, depending on the synthesis of new proteins or by stabilizing existing proteins.
4. References
[1] Watanabe-Asano T, Kuma A, Mizushima N. Cycloheximide inhibits starvation-induced autophagy through mTORC1 activation. Biochem Biophys Res Commun. 2014 Mar 7;445(2):334-9. doi: 10.1016/j.bbrc.2014.01.180. Epub 2014 Feb 10. PMID: 24525133.
[2] Al Nebaihi HM, Davies NM, Brocks DR. Pharmacokinetics of cycloheximide in rats and evaluation of its effect as a blocker of intestinal lymph formation. Eur J Pharm Biopharm. 2023 Dec;193:89-95. doi: 10.1016/j.ejpb.2023.10.016. Epub 2023 Oct 24. PMID: 37884159.
[3] Zhao Q, Bolton B, Rothe R, et al. Cycloheximide-resistant ribosomes reveal adaptive translation dynamics in C. elegans. Genetics. 2026 Jan 7;232(1):iyaf189. doi: 10.1093/genetics/iyaf189. PMID: 40929375; PMCID: PMC12477835.
[4] Xu Q, Tan B, Lin P, Wang Y, Huang D, Yang X, Lu J, Chen D, Hu J, Li L, Wu M. Salvianolic acid C protects against sepsis-associated acute kidney injury through promotion of PGC1α-mediated renal gluconeogenesis. Phytomedicine. 2026 Feb;151:157796. doi: 10.1016/j.phymed.2026.157796. Epub 2026 Jan 8. PMID: 41539097.
Other Articles
Subscription to TargetMol News
An essential round-up of science news, opinion and analysis, delivered to your inbox every weekday.

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.