Your shopping cart is currently empty
Your shopping cart is currently empty
TargetMol—Alkaloid—Chloroquine (Cat. No. T8689, CAS 54-05-7), a Classic Lysosomal Regulator
1. Product Introduction
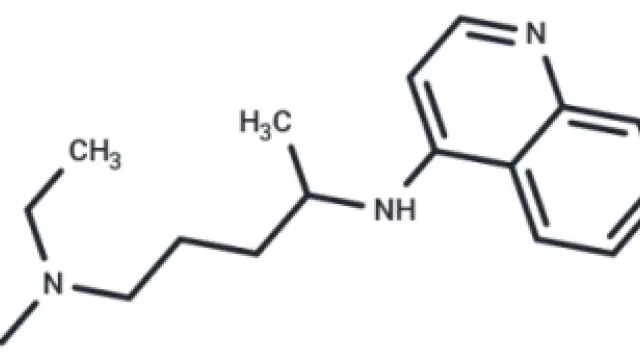
Chloroquine (Cat. No. T8689, CAS 54-05-7), also known as CQ. Chloroquine is a Toll-like receptor inhibitor that inhibits autophagy. Chloroquine has anti-malarial and anti-inflammatory activities and is widely used in the treatment of malaria and rheumatoid arthritis. Chloroquine also has anti-SARS-CoV-2 (COVID-19) activity and anti-HIV-1 activity.
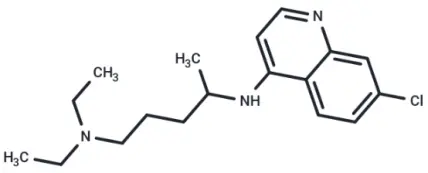
Molecular structure of Chloroquine
2. Background Introduction
Chloroquine (CQ) is a classic anti-malarial drug. Its role is not limited to anti-parasites, but also involves a variety of intracellular signaling pathways. As a weakly basic small molecule, chloroquine can be protonated and accumulated in acidic organelles such as endosomes and lysosomes, thereby inhibiting the acidic environment and function of these organelles. This property makes it a commonly used autophagy inhibitor in experiments. In addition, chloroquine can interfere with a variety of Toll-like receptors (TLRs) signal transduction, such as inhibiting the activation of TLR7/9, which helps regulate the inflammatory response and immune response. Chloroquine is therefore described as a multi-target drug with autophagy inhibition and TLR inhibition.
Chloroquine has a variety of clinical and experimental uses due to its broad-spectrum biological effects. It was first used as an effective anti-malarial drug for the treatment of Plasmodium infection, and was later found to have significant anti-inflammatory activity, so it was also used to treat autoimmune diseases such as rheumatoid arthritis. Chloroquine has shown inhibitory effects on a variety of viruses in vitro experiments, including infection models such as SARS-CoV-2 (triggering COVID-19) and HIV-1. Its antiviral effect may be related to mechanisms such as increasing endosomal/lysosomal pH, inhibiting the binding of virus to cell receptors, and interfering with vesicle transport after virus invasion, although the efficacy of these mechanisms in vivo still needs to be further verified. [1]
3. Application References
Pulmonary Delivery of Aerosolized Chloroquine and Hydroxychloroquine to Treat COVID-19: In Vitro Experimentation to Human Dosing Predictions

Research Overview:
In this study, the pulmonary aerosol delivery strategy of chloroquine and hydroxychloroquine was systematically evaluated in vitro. Primary human bronchial epithelial cells were used to explore the feasibility of increasing local exposure of drugs in lung tissue and reducing systemic toxicity by aerosol inhalation. Firstly, an in vitro aerosol generation and deposition model was constructed to determine the physical and chemical stability, particle size distribution and lung deposition efficiency of the two drugs during atomization. Then, combined with the anti-SARS-CoV-2 activity data at the cellular level, a pharmacokinetic model from in vitro experiments to human lung dose prediction was established. The results showed that compared with oral administration, aerosol delivery could achieve higher effective drug concentration in the lungs and significantly reduce the risk of systemic circulatory exposure. This study provides an experimental basis and dose estimation framework for the local administration strategy of chloroquine / hydroxychloroquine in respiratory tract infection by combining in vitro pharmacodynamic experiments, delivery engineering and pharmacokinetic modeling. [2]
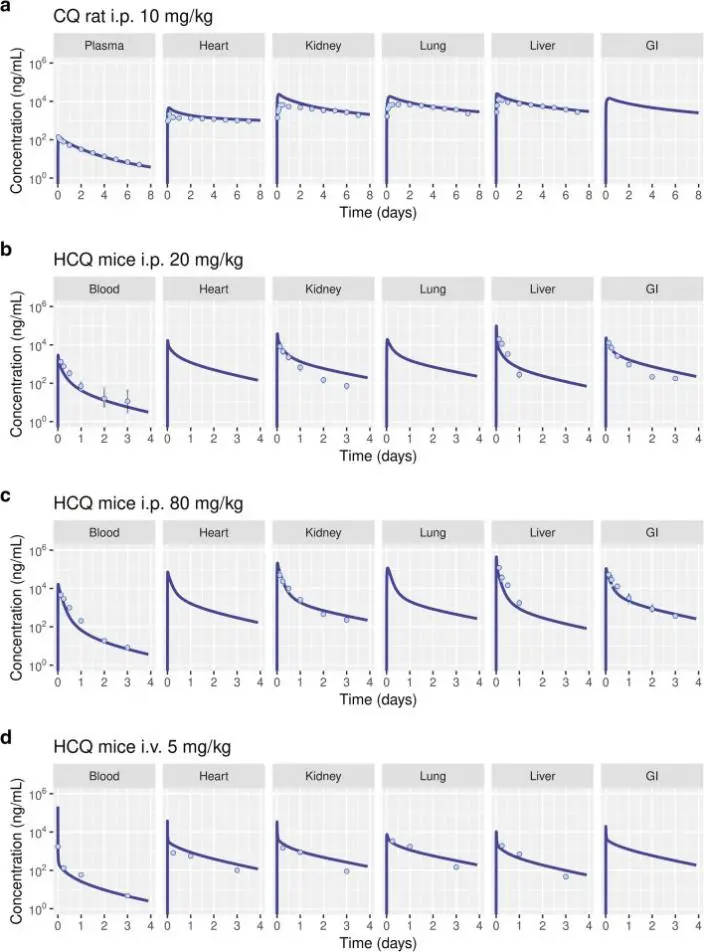
Concentration-time curve predicted by a physiologically based pharmacokinetic model [2]
Chloroquine ameliorates carbon tetrachloride-induced acute liver injury in mice via the concomitant inhibition of inflammation and induction of apoptosis

Research Overview:
In this study, the hepatoprotective effect of chloroquine was evaluated in a mouse model of CCl4-induced acute liver injury. Through serum transaminase, liver histopathology and molecular biology detection, it was found that chloroquine treatment significantly reduced carbon tetrachloride-induced hepatocyte necrosis and inflammatory infiltration. Mechanistically, on the one hand, chloroquine down-regulates the expression of pro-inflammatory factors and inhibits the activation of inflammatory signaling pathways. On the other hand, it promotes the programmed apoptosis rather than necrosis of damaged hepatocytes, thereby reducing the secondary inflammatory amplification effect. This study shows that chloroquine improves the pathological process of acute toxic liver injury through the synergistic regulation of 'anti-inflammatory + pro-apoptotic', and provides an in vivo experimental basis for its potential reuse in liver injury intervention. [3]
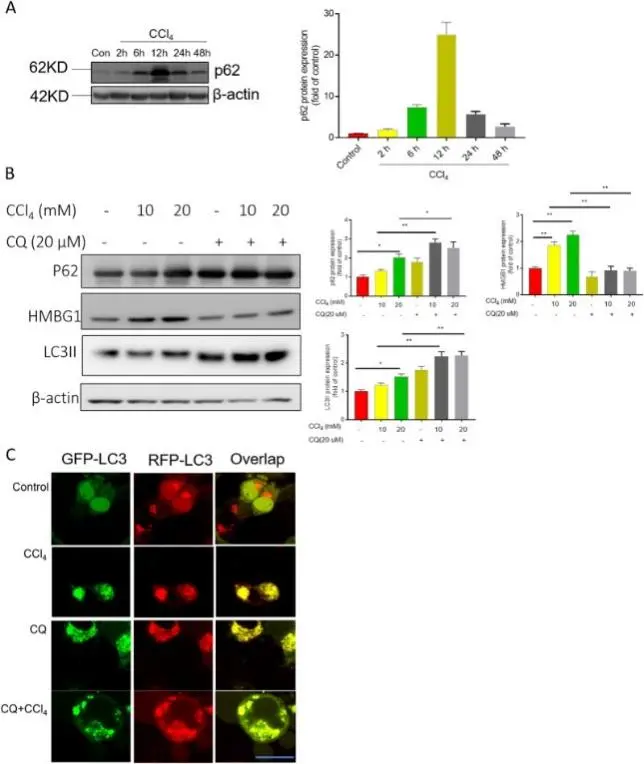
CCl4 blocks autophagy flux [3]
Modulation of O-GlcNAc cycling influences α-synuclein amplification, degradation, and associated neuroinflammatory pathology

This study systematically explored the effects of regulating O-GlcNAc cycle (controlled by O-GlcNAc transferase OGT and O-GlcNAcase OGA) on the aggregation, transmission, degradation and related neuroinflammation of Parkinson 's disease-related protein α-synuclein (α-syn). They found in cell and mouse models that the transmission of pathological α-syn reduced O-GlcNAc levels and OGT expression in recipient cells, while inhibition of OGT aggravated α-syn aggregation, cross-cellular transmission and NLRP3-mediated microglial inflammation. In contrast, inhibition of OGA to increase O-GlcNAc modification can alleviate these pathological changes and promote the clearance of α-syn aggregates through the autophagy-lysosomal pathway. These results indicate that the regulation of O-GlcNAc modification can affect the pathological process of α-syn, and provide a mechanism basis for the treatment of α-syn disease by changing this post-translational modification. [4]
In this article, Chloroquine was used as a pharmacological tool to inhibit the autophagic lysosomal pathway, together with another autophagy inhibitor 3-MA, to verify the mechanism by which O-GlcNAc modification promotes the degradation of pathological α-synuclein (α-syn) through the autophagic-lysosomal pathway.
4. References
[1] Al-Hamadani M, Darweesh M, Mohammadi S, et al. Chloroquine and hydroxychloroquine: Immunomodulatory effects in autoimmune diseases. World J Biol Chem. 2025 Jun 5;16(2):107042. doi: 10.4331/wjbc.v16.i2.107042. PMID: 40476257; PMCID: PMC12136085.
[2] Kolli AR, Semren TZ, Bovard D, et al. Pulmonary Delivery of Aerosolized Chloroquine and Hydroxychloroquine to Treat COVID-19: In Vitro Experimentation to Human Dosing Predictions. AAPS J. 2022 Feb 7;24(1):33. doi: 10.1208/s12248-021-00666-x. PMID: 35132508; PMCID: PMC8821864.
[3] Dai C, Xiao X, Li D, et al. Chloroquine ameliorates carbon tetrachloride-induced acute liver injury in mice via the concomitant inhibition of inflammation and induction of apoptosis. Cell Death Dis. 2018 Nov 26;9(12):1164. doi: 10.1038/s41419-018-1136-2. PMID: 30478280; PMCID: PMC6255886.
[4] Miao Y, Zhang T, Ma Z, Du H, Gu Q, Jiang M, Xiong K, Liu CF, Meng H. Modulation of O-GlcNAc cycling influences α-synuclein amplification, degradation, and associated neuroinflammatory pathology. Mol Neurodegener. 2025 Oct 27;20(1):113. doi: 10.1186/s13024-025-00904-2. PMID: 41146299; PMCID: PMC12560605.
Other Articles
Subscription to TargetMol News
An essential round-up of science news, opinion and analysis, delivered to your inbox every weekday.

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.