Your shopping cart is currently empty
Your shopping cart is currently empty
TargetMol—Star Molecules—Erastin (Cat. No. T1765, Cas. 571203-78-6), Targeting Mitochondrial VDAC
1. Product Introduction
Erastin (Cat. No. T1765, Cas. 571203-78-6), is a ferroptosis activator targeting mitochondrial VDAC, acting in a ROS- and iron-dependent manner. Erastin exhibits antitumor activity, selectively targeting tumor cells harboring oncogenic RAS mutations.
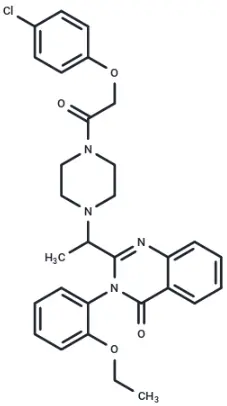
Molecular structure of Erastin
2. Background Introduction
The voltage-dependent anion channel (VDAC) is the primary channel protein on the mitochondrial outer membrane, responsible for the exchange of small metabolites, ions, and respiratory substrates between mitochondria and the cytosol. It is crucial for maintaining mitochondrial membrane potential, energy metabolism, and redox homeostasis. VDAC has three major isoforms (VDAC1–3), among which VDAC2/3 have been found to participate in regulating oxidative stress and non-apoptotic programmed cell death pathways such as ferroptosis. VDAC channels can also interact with cellular components, including microtubules, to modulate mitochondrial metabolic states and ROS production, forming a key basis for metabolic reprogramming in tumor cells. Since tumor cells often exhibit high ROS burden and are particularly sensitive to mitochondrial dysfunction, VDAC serves as a central hub linking metabolism and death signaling, making it a highly attractive therapeutic target for ferroptosis induction strategies. [1]
Erastin is one of the earliest identified ferroptosis activators and disrupts cellular homeostasis through multiple mechanisms. On one hand, it directly binds to and modulates the function of VDAC2/3 channels, altering mitochondrial substrate exchange and energy metabolism. On the other hand, Erastin inhibits the activity of the cystine–glutamate antiporter on the plasma membrane, leading to impaired cysteine uptake and decreased intracellular glutathione (GSH) levels, thereby promoting iron-dependent lipid peroxidation and ROS accumulation. The dual dependence on ROS and iron is a hallmark of ferroptosis, with the accumulation of lipid peroxides in the cell membrane ultimately resulting in cell death. Erastin shows pronounced selectivity for tumor cells harboring oncogenic RAS mutations, likely due to their unique reliance on redox homeostasis and metabolic regulation, which underlies its potential antitumor activity in these models. [1]
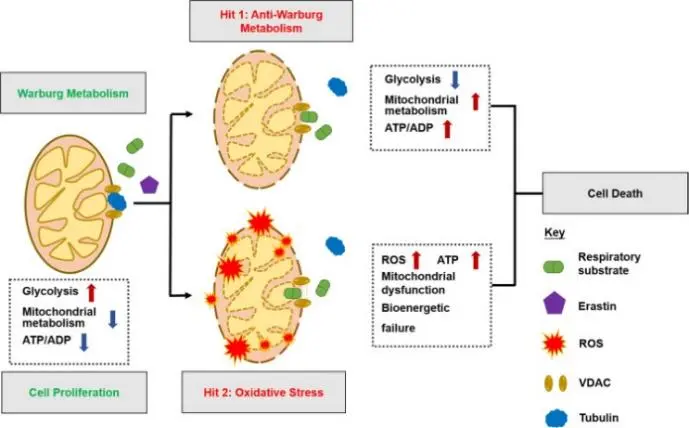
Erastin induces ferroptosis by altering the permeability of VDAC [1]
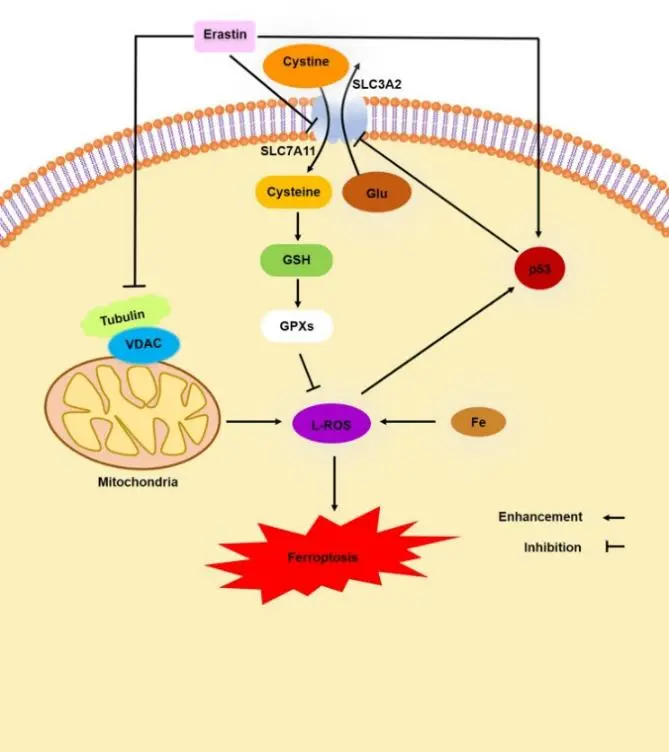
Erastin-induced ferroptosis-associated pathways [1]
3. Application References
Nedd4 ubiquitylates VDAC2/3 to suppress erastin-induced ferroptosis in melanoma

Research Overview:
This study uncovers a novel regulatory mechanism of ferroptosis in melanoma cells: the E3 ubiquitin ligase Nedd4 ubiquitinates the mitochondrial outer membrane proteins VDAC2/3, reducing their stability and functional activity, thereby suppressing Erastin-induced ferroptosis. In melanoma models, high Nedd4 expression significantly attenuates Erastin-triggered ROS accumulation and lipid peroxidation, enhancing cellular resistance to ferroptosis. Conversely, inhibition or knockdown of Nedd4 restores VDAC2/3 function, rendering cells more sensitive to Erastin. By elucidating the molecular mechanism underlying cellular resistance to Erastin, the study reveals how cells adapt via novel molecules to maintain homeostasis. Moreover, the FOXM1–Nedd4–VDAC2/3 negative feedback loop mediating Erastin resistance provides a preliminary framework for overcoming resistance to ferroptosis inducers. [2]
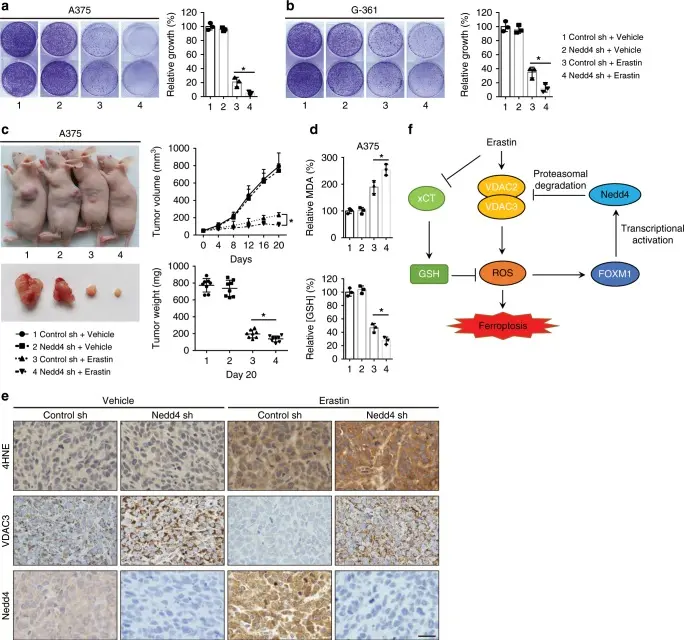
Knockdown of Nedd4 suppresses Erastin resistance both in vitro and in vivo [2]
Melatonin and erastin emerge synergistic anti-tumor effects on oral squamous cell carcinoma by inducing apoptosis, ferroptosis, and inhibiting autophagy through promoting ROS

Research Overview:
This study found that in oral squamous cell carcinoma models, combined treatment with Melatonin and Erastin produces a significant synergistic antitumor effect. Mechanistically, Melatonin enhances Erastin-induced intracellular ROS accumulation, further amplifying iron-dependent lipid peroxidation stress and thereby strengthening ferroptosis signaling. Simultaneously, the combination synergistically activates mitochondria-mediated apoptotic pathways while inhibiting protective autophagy, disrupting redox and metabolic homeostasis in tumor cells. The results indicate that co-treatment with Melatonin and Erastin regulates ROS to activate multiple forms of regulated cell death (RCD), including apoptosis, autophagy, and ferroptosis, demonstrating a synergistic antitumor effect in oral cancer. Novel strategies targeting the crosstalk between different RCD forms hold promise for improving anticancer efficacy and overcoming resistance. [3]
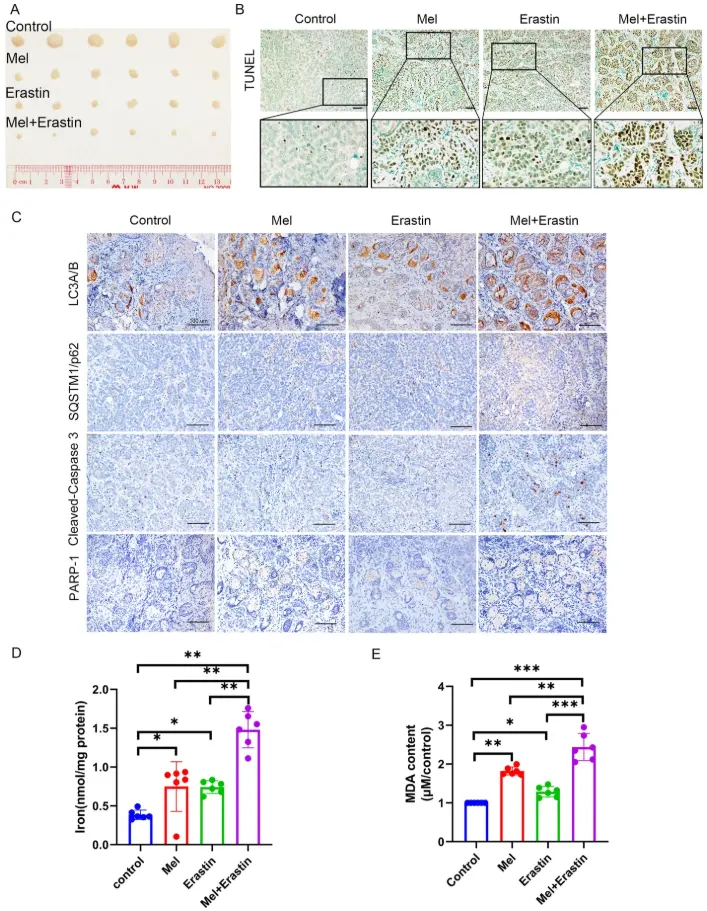
Effects of Melatonin combined with Erastin on apoptosis, autophagy and ferroptosis in vivo [3]
1.%2. AAK1 activation-mediated iron trafficking drives ferroptotic cell death

Research Overview:
This study revealed a new mechanism regulating cell iron metabolism and ferroptosis: during ferroptosis induction, PKCβII kinase phosphorylates and activates AAK1, which in turn promotes the phosphorylation of AP2M1 by AAK1 and promotes clathrin-mediated endocytosis of TFR1 receptors, thereby increasing cellular iron uptake and available divalent iron levels. This increase in iron supply promotes lipid peroxidation and ferroptosis, blocking the activation of AAK1 can inhibit ferroptosis and promote breast tumor growth in vivo, suggesting that this signaling pathway is not only the core mechanism of ferroptosis regulation, but also has potential therapeutic significance. [4]
In this study, Erastin was used as a classic ferroptosis inducer to trigger iron-dependent lipid peroxidation and ferroptosis in cells, thereby serving as a functional tool to verify the key role of AAK1-mediated iron transport in ferroptosis. The researchers observed that AAK1 activation can significantly enhance Erastin-induced iron uptake and lipid peroxidation levels by Erastin-treated cells. Inhibition of AAK1 or blocking its downstream signals can significantly attenuate Erastin-induced ferroptosis, demonstrating that the AAK1-AP2M1-TFR1 axis plays a central regulatory role in Erastin-triggered ferroptosis.
4. References
[1] Zhao Y, Li Y, Zhang R, et al. The Role of Erastin in Ferroptosis and Its Prospects in Cancer Therapy. Onco Targets Ther. 2020 Jun 11,13:5429-5441. doi: 10.2147/OTT.S254995. PMID: 32606760, PMCID: PMC7295539.
[2] Yang Y, Luo M, Zhang K, et al. Nedd4 ubiquitylates VDAC2/3 to suppress erastin-induced ferroptosis in melanoma. Nat Commun. 2020 Jan 23,11(1):433. doi: 10.1038/s41467-020-14324-x. PMID: 31974380, PMCID: PMC6978386.
[3] Wang L, Wang C, Li X, et al. Melatonin and erastin emerge synergistic anti-tumor effects on oral squamous cell carcinoma by inducing apoptosis, ferroptosis, and inhibiting autophagy through promoting ROS. Cell Mol Biol Lett. 2023 May 2,28(1):36. doi: 10.1186/s11658-023-00449-6. PMID: 37131152, PMCID: PMC10155313.
[4] Li LC, Ye ZP, Xiao Y, et al. AAK1 activation-mediated iron trafficking drives ferroptotic cell death. Nat Commun. 2025 Dec 17,17(1):819. doi: 10.1038/s41467-025-67523-9. PMID: 41407700, PMCID: PMC12824188.
Other Articles
Subscription to TargetMol News
An essential round-up of science news, opinion and analysis, delivered to your inbox every weekday.

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.