Your shopping cart is currently empty
Your shopping cart is currently empty
TargetMol—Star Molecules—Daraxonrasib & RMC-7977 & Zoldonrasib, RMC Series Inhibitory Effect on RAS Models
Daraxonrasib (RMC-6236), RMC-7977, and Zoldonrasib (RMC-9805) were all developed on the RAS(ON) tri-complex inhibitor platform pioneered by Revolution Medicines. This class of compounds leverages an induced-proximity mechanism to recruit oncogenic RAS proteins to the intracellular chaperone Cyclophilin A, thereby reshaping the surface topology of Cyclophilin A to form a high-affinity ternary complex that selectively recognizes the GTP-bound (active) state of RAS. This mechanism effectively creates a physical blockade that prevents RAS from interacting with downstream effectors such as RAF, thereby shutting down signal transduction. While the three compounds share a highly unified molecular mechanism, their differentiation lies primarily in target spectrum selectivity (e.g., RMC-6236 exhibits pan-RAS inhibitory activity across multiple RAS variants), enhanced chemical engagement strategies (e.g., RMC-9805 incorporates covalent modification to improve targeting durability toward KRASG12D), and distinct R&D positioning and functional roles (e.g., RMC-7977 serves as a mechanistic probe molecule to systematically characterize the coverage and biochemical effects of non-covalent ternary complex formation across multiple RAS(ON) subtypes).
1. Product Introduction
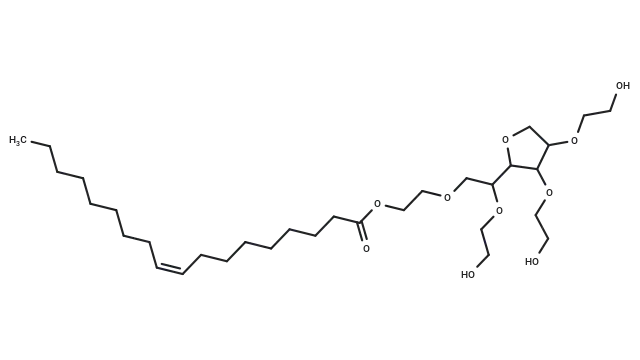
Daraxonrasib (Cat. No. T74698, CAS. 2765081-21-6), also known as RMC-6236, is an orally active, novel tri-complex RAS(ON) multi-selective inhibitor. It is a potent non-covalent inhibitor that targets the GTP-bound state of multiple RAS variants. Daraxonrasib exhibits antitumor activity and can be used in research related to RAS-driven cancers.
Molecular structure of Daraxonrasib
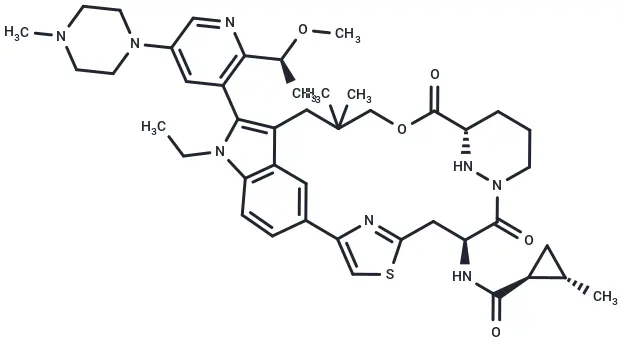
RMC-7977 (Cat. No. T81263, CAS. 2765082-12-8) is a highly selective inhibitor of the active (GTP-bound) forms of KRAS, HRAS, and NRAS. It exhibits anticancer activity and can be used for research on solid tumors harboring the KRASG12C mutation.
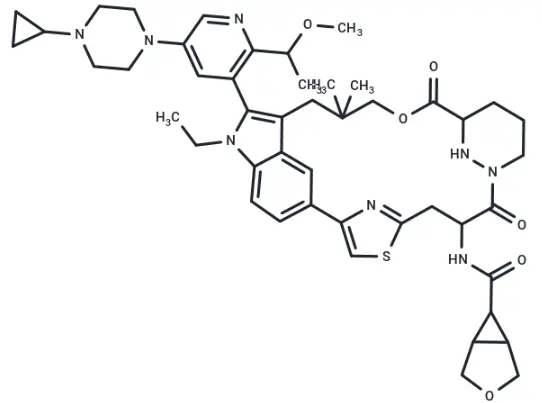
Molecular structure of RMC-7977
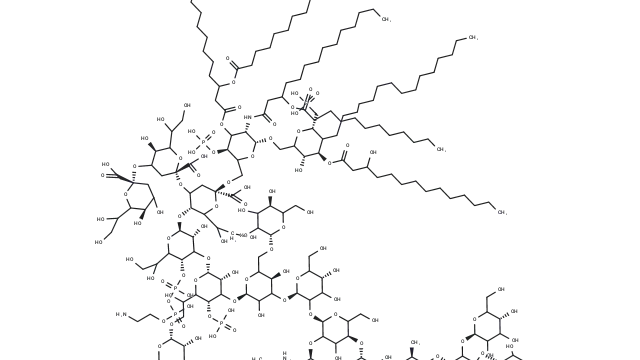
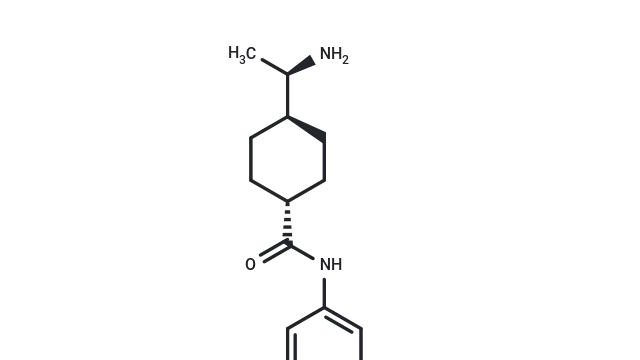
Zoldonrasib (Cat. No. T78212, CAS. 2922732-54-3), also known as RMC-9805, is a novel, mutation-selective, covalent, orally active KRASG12D(ON) inhibitor. It forms a stable, high-affinity tri-complex with KRASG12D and Cyclophilin A, thereby disrupting interactions with downstream effectors and inhibiting KRASG12D(ON) signaling. RMC-9805 induces apoptosis and promotes tumor regression in preclinical models of KRASG12D-driven tumors.
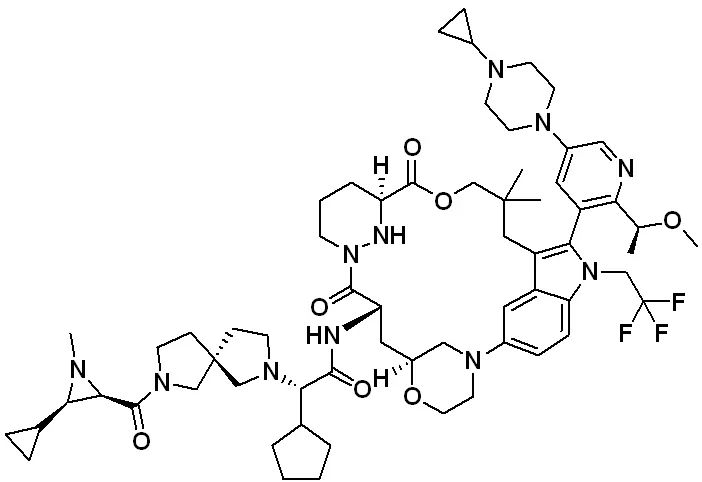
Molecular structure of RMC-9805
2. Background Introduction
RAS proteins—including the three isoforms KRAS, NRAS, and HRAS—are among the most common oncogenic drivers in human cancers, with approximately one-third of malignancies associated with RAS mutations. Under physiological conditions, RAS functions as a molecular switch, cycling between a GDP-bound “OFF” state and a GTP-bound “ON” state. Oncogenic mutations lock RAS in the GTP-bound state, leading to persistent activation of downstream signaling pathways (such as MAPK and PI3K), which in turn drives abnormal cell proliferation and tumor growth.
Because the surface of RAS is relatively smooth and lacks conventional “druggable” pockets for small-molecule binding, it has long been considered an “undruggable” target. Therefore, developing inhibitors that can directly target the active, GTP-bound state of RAS is of great significance.
Daraxonrasib is a non-covalent molecule with broad inhibitory activity across RAS isoforms and mutants, enabling it to simultaneously target multiple common RAS-driven tumors, including those harboring KRAS, NRAS, and HRAS mutations. Daraxonrasib forms a ternary complex with GTP-bound RAS and the chaperone protein Cyclophilin A, thereby blocking downstream RAS signaling and inhibiting tumor cell proliferation. This inhibitor exhibits broad activity against multiple RAS mutants as well as wild-type RAS. [1]
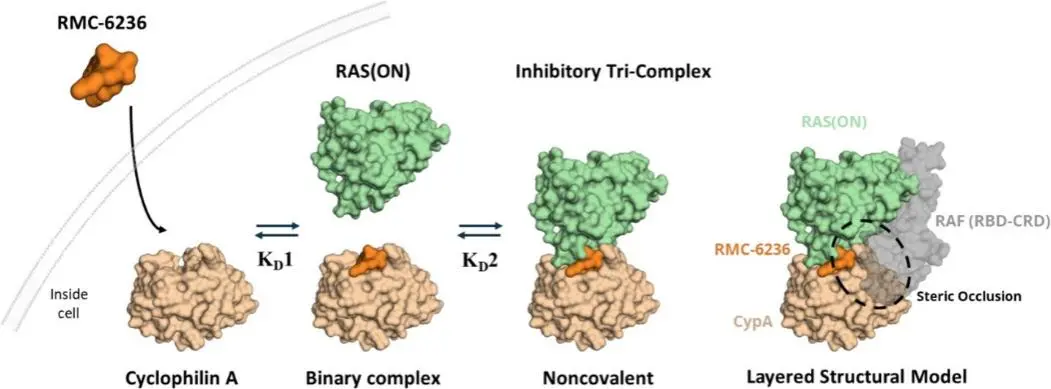
The tri-complex platform enables selective targeting of oncogenic RAS(ON) proteins [1]
RMC-7977 is a reversible, tri-complex RAS inhibitor with broad activity against the active forms of mutant and wild-type KRAS, NRAS, and HRAS, functioning as a multi-selective RAS(ON) inhibitor. In preclinical studies, RMC-7977 demonstrated potent inhibitory effects in RAS-dependent tumor models harboring diverse RAS genotypes, with particularly strong efficacy in cancer models containing codon 12 KRAS mutations (KRASG12X). Treatment with RMC-7977 induced tumor regression across multiple RAS-dependent preclinical tumor models and was well tolerated. Moreover, RMC-7977 retained significant growth-inhibitory activity in KRASG12X cancer models that had developed resistance to KRASG12X inhibitors due to reactivation of RAS pathway signaling. [2]
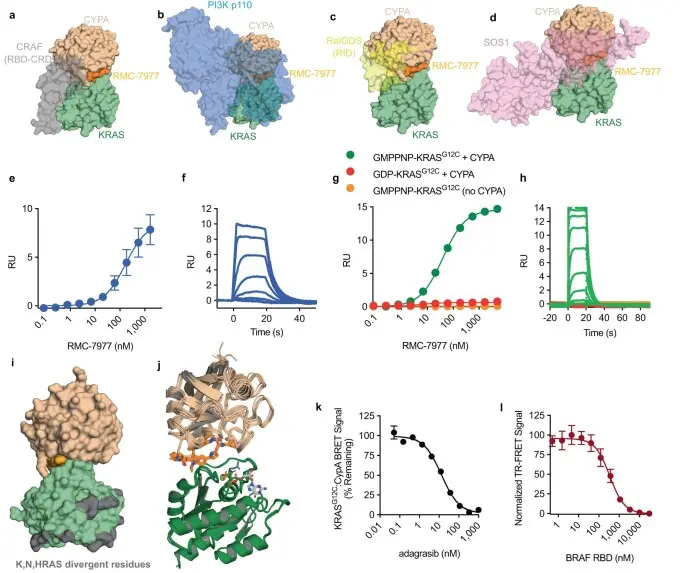
RMC-7977 inhibits the active states of multiple RAS variants [2]
Zoldonrasib is a novel covalent inhibitor that promotes the formation of a stable, high-affinity tri-complex between Cyclophilin A and GTP-bound KRASG12X, thereby suppressing KRASG12X(ON) downstream signaling by sterically blocking the binding of effector proteins such as RAF, PI3K, and RALGDS. More than 90% of pancreatic ductal adenocarcinoma (PDAC) cases are driven by oncogenic KRAS mutations. KRAS mutations—particularly the predominant G12D mutation in PDAC—promote tumor initiation, progression, and immune evasion, leading to therapeutic resistance. Due to the low intrinsic reactivity of the Asp12 residue, KRASG12X has long been considered an “undruggable” target. However, this molecular glue–based mechanism enables effective inhibition of the active KRASG12X state and has demonstrated significant antitumor activity across multiple preclinical models, offering a new strategy to overcome the therapeutic challenges associated with KRASG12X -driven malignancies. [3]
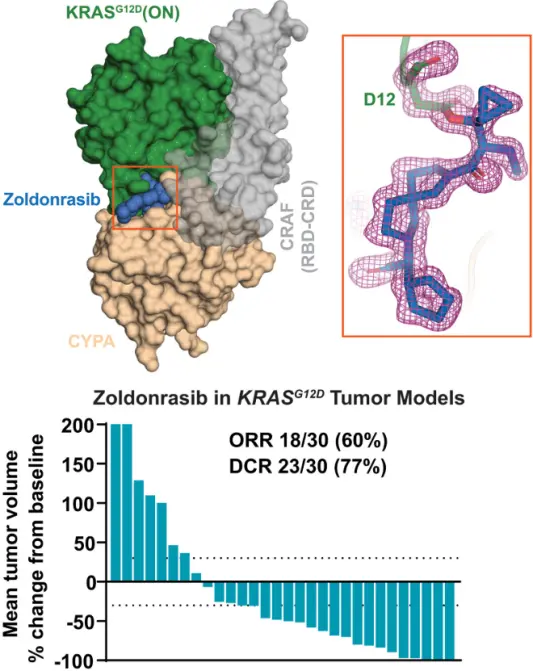
Zoldonrasib binds to Cyclophilin A and KRASG12D [3]
3. Application References
Translational and Therapeutic Evaluation of RAS-GTP Inhibition by RMC-6236 in RAS-Driven Cancers

Research Overview:
This study systematically evaluated the translational and therapeutic potential of Daraxonrasib as a multi-selective RAS(ON) inhibitor targeting the GTP-bound state across diverse RAS-driven cancers. A series of in vitro biochemical and cellular assays first confirmed the non-covalent binding of RMC-6236 to the GTP-bound forms of multiple RAS mutants—including KRAS, NRAS, and HRAS—and its ability to disrupt RAS–RAF interactions. Subsequent studies in multiple human tumor xenograft models demonstrated dose-dependent suppression of RAS signaling following oral administration, significant tumor volume regression, and durable inhibitory effects, providing initial evidence for the clinical feasibility and promise of this class of pan-RAS GTP-state inhibitors. [4]
In this study, in vitro experiments were conducted in which Daraxonrasib was used to treat HPAC and Capan-2 cells. Time- and concentration-dependent inhibition of RAS pathway signaling markers, including pERK, pAKT, and pS6, was observed. HPAC cells also exhibited sustained suppression of pERK and time-dependent induction of apoptosis. Among RAS-mutant cancer cell lines, those harboring KRASG12X and NRASQ61X mutations showed markedly higher sensitivity compared with cell lines carrying other oncogenic KRAS or NRAS mutations.
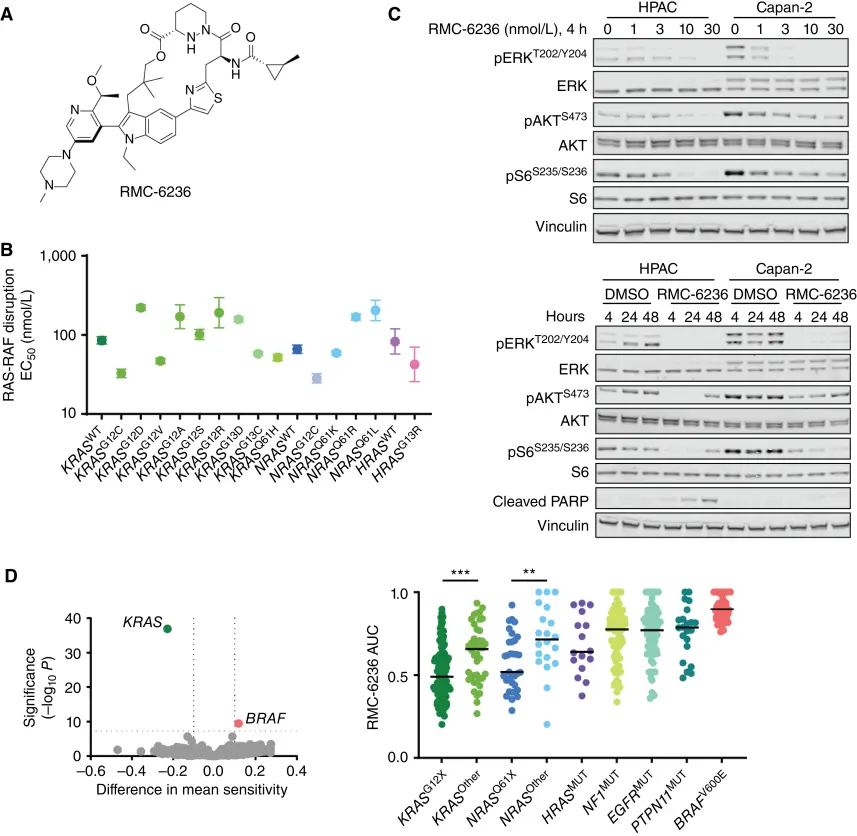
Daraxonrasib can inhibit the GTP-bound state of multiple RAS variants in vitro [4]
RAS(ON) Multiselective Inhibition Drives Antitumor Immunity in Preclinical Models of NRAS-Mutant Melanoma

Research Overview:
RMC-7977 is a preclinical tool compound used to validate the RAS(ON) tri-complex inhibition strategy and represents a class of orally active, multi-selective RAS(ON) inhibitors that includes the investigational candidate RMC-6236. RMC-7977 binds to and remodels the surface of Cyclophilin A, forming a high-affinity binary complex with active RAS [RAS(ON)]. This sterically blocks the interaction between RAS and downstream effector proteins, thereby inhibiting downstream signaling pathways. RMC-7977 has been shown to induce potent antitumor immune responses in multiple NRAS-mutant melanoma models. Following RMC-7977 treatment, rapid tumor regression can be triggered through inhibition of the MAPK pathway, upregulation of MHC and PD-L1 expression, and enhanced infiltration of CD4+ and CD8+ T cells. [5]
This study systematically elucidated the antitumor activity of RMC-7977 in NRAS-mutant melanoma mouse models. The results showed that the durable antitumor responses induced by RMC-7977 depended on the activation of CD4+ and CD8+ T cells and were accompanied by a reduction in the recruitment of myeloid-derived suppressor cells. Moreover, when combined with anti–PD-1 antibodies, RMC-7977 significantly prolonged survival and achieved complete tumor regression in preclinical models resistant to anti–PD-1 therapy.
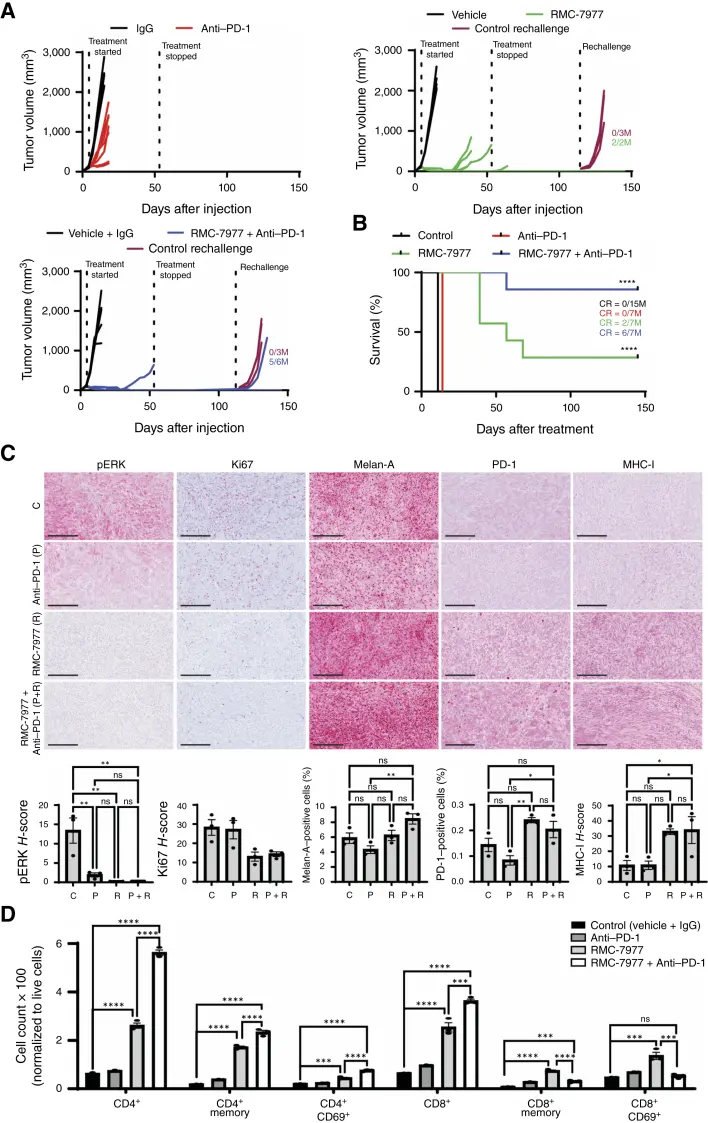
Combination therapy with anti–PD-1 treatment and RMC-7977 increases the rate of durable and complete tumor regression [5]
Concurrent inhibition of oncogenic and wild-type RAS-GTP for cancer therapy

Research Overview:
This study investigated RMC-7977, a RAS(ON) multi-selective inhibitor related to RMC-6236, and demonstrated its potent activity in “RAS-dependent” tumors harboring diverse RAS genotypes. Treatment with RMC-7977 induced tumor regression and showed good tolerability across multiple RAS-dependent preclinical cancer models. RMC-7977 also inhibited the growth of KRASG12C cancer models that had developed resistance to KRASG12C inhibitors due to reactivation of RAS pathway signaling. These findings suggest that RAS(ON) multi-selective inhibitors can simultaneously target multiple oncogenic and wild-type RAS isoforms, offering therapeutic potential for a broad range of RAS-dependent cancers with high unmet clinical needs. Meanwhile, RMC-6236 is currently undergoing clinical investigation in patients with KRAS-mutant solid tumors. [2]
This study performed cell viability profiling (PRISM assay) across 869 human tumor cell lines with diverse genetic backgrounds and histological subtypes to identify genetic features associated with sensitivity or resistance to RMC-7977. Oncogenic KRAS mutation status emerged as an important genetic marker of sensitivity to RMC-7977. At a dose of 10 mg/kg administered daily, RMC-7977 was well tolerated in the NCI-H441 model, producing an average tumor regression of 83% after 28 days of treatment.
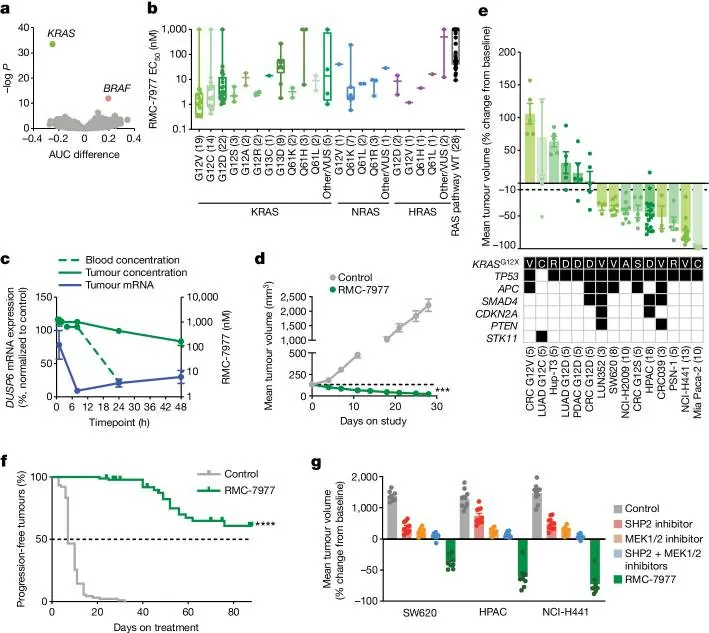
RMC-7977 is widely used in RAS-dependent cancer models [2]
A neomorphic protein interface catalyzes covalent inhibition of RASG12D aspartic acid in tumors

Research Overview:
This study developed a series of compounds that induce a protein–protein interaction between RAS and the cellular chaperone Cyclophilin A via a RAS inhibitor, thereby enabling selective targeting of KRASG12D. These compounds exploit the interface formed between the two proteins and, in a Cyclophilin A–dependent manner, catalytically achieve irreversible inhibition of the aspartate residue in mutant RAS. One representative inhibitor from this class is Zoldonrasib, which, following oral administration in mice, irreversibly modifies mutant RAS and significantly suppresses tumor growth in experimental models of lung, pancreatic, and colorectal cancers. [3]
This study showed that, in the HPAC xenograft model, daily oral administration of Zoldonrasib achieved sufficient in vivo exposure and stable covalent target engagement with KRASG12D, leading to inhibition of the RAS/MAPK signaling pathway. Zoldonrasib was well tolerated in the model and exhibited marked dose-dependent antitumor activity, inducing up to 93% durable tumor regression.
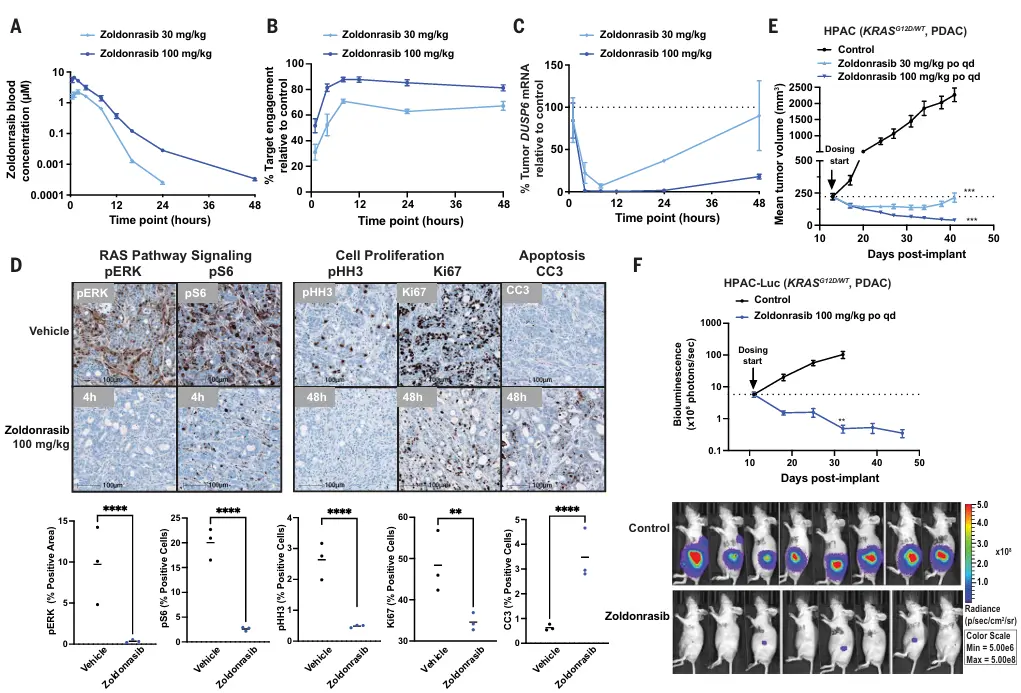
KRASG12D inhibitor Zoldonrasib blocks RAS signaling and induces tumor regression [3]
4. References
[1] Cregg J, Edwards AV, Chang S, et al. Discovery of Daraxonrasib (RMC-6236), a Potent and Orally Bioavailable RAS(ON) Multi-selective, Noncovalent Tri-complex Inhibitor for the Treatment of Patients with Multiple RAS-Addicted Cancers. J Med Chem. 2025 Mar 27;68(6):6064-6083. doi: 10.1021/acs.jmedchem.4c02314. Epub 2025 Mar 8. PMID: 40056080.
[2] Holderfield M, Lee BJ, Jiang J, et al. Concurrent inhibition of oncogenic and wild-type RAS-GTP for cancer therapy. Nature. 2024 May;629(8013):919-926. doi: 10.1038/s41586-024-07205-6. Epub 2024 Apr 8. PMID: 38589574; PMCID: PMC11111408.
[3] Weller C, Burnett GL, Jiang L, et al. A neomorphic protein interface catalyzes covalent inhibition of RASG12D aspartic acid in tumors. Science. 2025 Jul 24;389(6758):eads0239. doi: 10.1126/science.ads0239. Epub 2025 Jul 24. PMID: 40705880.
[4] Jiang J, Jiang L, Maldonato BJ, et al. Translational and Therapeutic Evaluation of RAS-GTP Inhibition by RMC-6236 in RAS-Driven Cancers. Cancer Discov. 2024 Jun 3;14(6):994-1017. doi: 10.1158/2159-8290.CD-24-0027. Erratum in: Cancer Discov. 2025 Oct 6;15(10):2186. doi: 10.1158/2159-8290.CD-25-1519. PMID: 38593348; PMCID: PMC11149917.
[5] Anastacio Da Costa Carvalho L, Tovbis Shifrin N, Phadke MS, et al. RAS(ON) Multiselective Inhibition Drives Antitumor Immunity in Preclinical Models of NRAS-Mutant Melanoma. Cancer Immunol Res. 2026 Jan 8;14(1):90-106. doi: 10.1158/2326-6066.CIR-25-0744. PMID: 41186497; PMCID: PMC12666865.
Other Articles
Subscription to TargetMol News
An essential round-up of science news, opinion and analysis, delivered to your inbox every weekday.

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.