Your shopping cart is currently empty
Your shopping cart is currently empty
TargetMol—Star Molecules—Trastuzumab (Cat. No. T9912, CAS. 180288-69-1), Inhibits HER2 Shedding
1. Product Introduction
Trastuzumab (Cat. No. T9912, CAS. 180288-69-1), also known as trastuzumab, is a humanized monoclonal antibody that selectively binds to HER2 with high affinity. Trastuzumab exhibits antitumor activity and is used for the treatment of HER2-positive tumors. It can induce cell cycle arrest, mediate cytotoxicity, and inhibit DNA damage repair.
2. Background Introduction
Aberrant alterations of HER2 (human epidermal growth factor receptor 2, also known as neu or erbB2) are closely associated with the initiation and progression of multiple cancers. HER2 has therefore become a key therapeutic target in oncology. Advances in molecular biology and deeper understanding of HER2-mediated signaling pathways have driven the development of drug design strategies and combination therapies. Current HER2-targeted therapeutics are broadly classified into antibodies, tyrosine kinase inhibitors (TKIs), and antibody–drug conjugates (ADCs), as illustrated in the figure. [1]
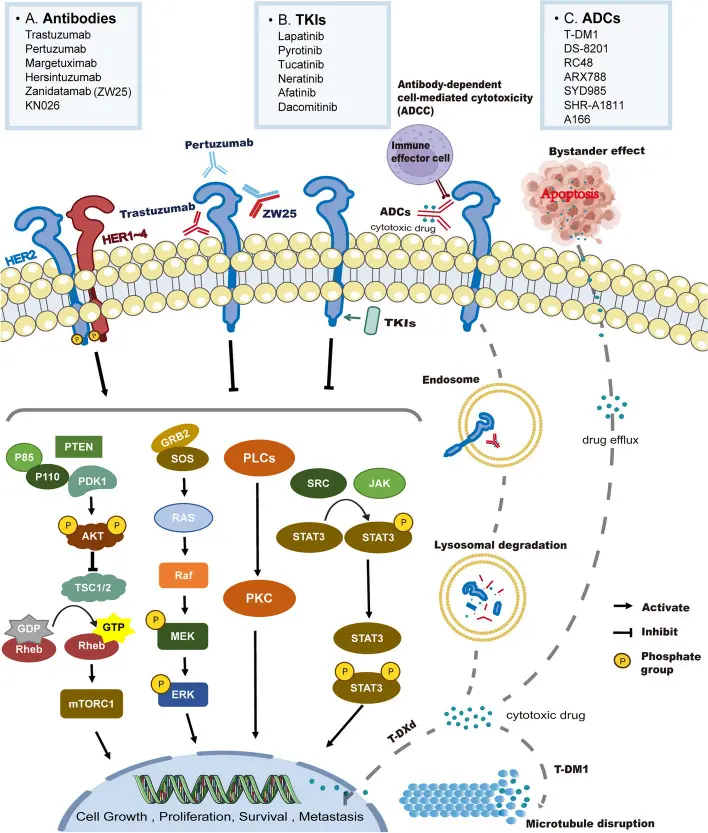
Classification and mechanisms of common anti-HER2 targeted therapies [1]
The antitumor activity of Trastuzumab is not mediated by a single pathway, but rather through the coordinated action of multiple mechanisms, including signaling pathway inhibition, regulation of receptor fate, and activation of immune effector functions. As shown in the figure, these mechanisms can be summarized into the following five aspects. [2]
(1) After binding to the HER2 receptor, Trastuzumab inhibits ligand-independent formation of HER2–HER3 heterodimers, HER3 phosphorylation, and the subsequent association of p85 with the HER3 receptor. This suppresses formation of the PI3K complex and thereby inhibits AKT phosphorylation. In addition, Trastuzumab inhibits AKT phosphorylation by activating PTEN phosphatase activity. Suppression of AKT phosphorylation leads to activation of p27KIP1, which translocates into the nucleus and induces cell cycle arrest by inhibiting the CDK2/cyclin E complex.
(2) Trastuzumab promotes HER2 homodimer formation and upregulates ADAM17 expression, thereby increasing the production of HER2-specific ligands and enhancing HER2 phosphorylation. The elevated levels of HER2-specific ligands derived from precursor forms further promote HER2 heterodimer formation.
(3) Binding of Trastuzumab to HER2 induces receptor internalization. Internalized HER2 can either be degraded or recycled back to the cell membrane.
(4) Trastuzumab induces dissociation of Shc from HER2 homodimers through a mechanism that is not yet fully understood, ultimately leading to inhibition of ERK1/2 phosphorylation.
(5) Trastuzumab induces antibody-dependent cellular cytotoxicity (ADCC). The Fc region of Trastuzumab is recognized by Fcγ receptors (FcγRs) on immune effector cells such as NK cells. Binding of the antibody Fc region to FcγRs activates downstream signaling in immune effector cells, leading to the release of perforin and granzymes. Granzymes then induce apoptosis in targeted tumor cells.
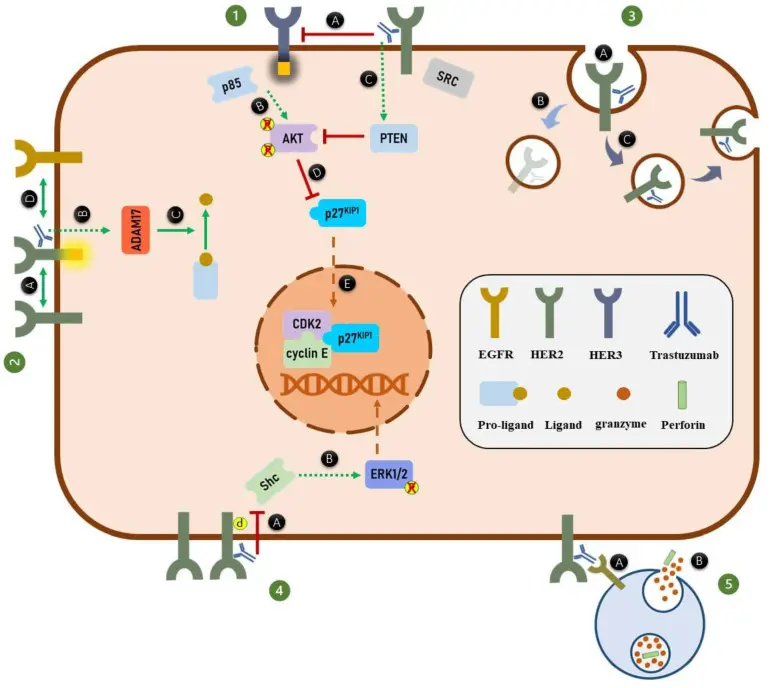
Mechanisms of action of Trastuzumab [2]
3. Application References
Trastuzumab, a monoclonal anti-HER2 antibody modulates cytotoxicity against cholangiocarcinoma via multiple mechanisms

Research Overview:
This study investigated the pharmacological effects of Trastuzumab in cholangiocarcinoma (CCA) models, demonstrating its substantial potential to expand from traditionally HER2-overexpressing tumors into the setting of HER2-low expression. The results showed that even in liver fluke–associated CCA models with low receptor expression, Trastuzumab not only inhibited tumor growth through direct suppression of signaling pathways but also exerted antitumor effects by activating multiple immune mechanisms. Through NK cell–mediated antibody-dependent cellular cytotoxicity (ADCC), macrophage-mediated antibody-dependent cellular phagocytosis (ADCP), and complement-dependent cytotoxicity (CDC), Trastuzumab displayed significant antitumor activity both in vitro and in vivo, including CDX and PDX models. These findings redefine the value of HER2 as an effective therapeutic target in cholangiocarcinoma and support the use of Trastuzumab as a potent antibody-based immunotherapy to address the substantial unmet clinical needs in CCA treatment. [3]
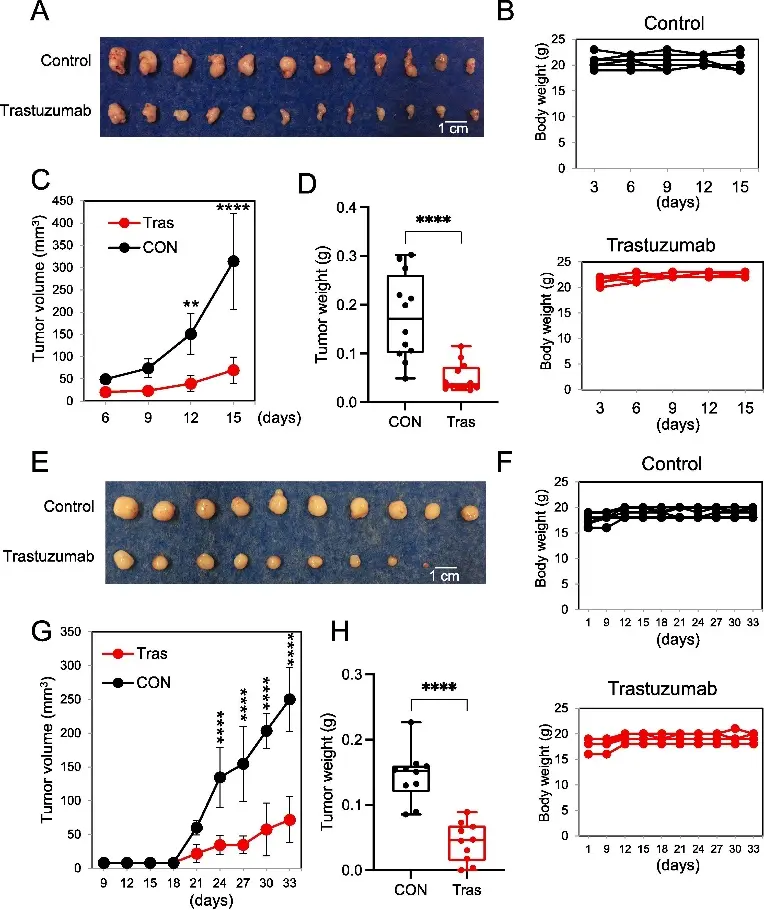
Trastuzumab inhibits tumor growth in both CCA xenograft and PDX models [3]
CMTM6 overexpression confers trastuzumab resistance in HER2-positive breast cancer

Research Overview:
This study showed that CMTM6 directly interacts with HER2 and inhibits its ubiquitin-mediated degradation, thereby stabilizing HER2 protein levels, enhancing HER2 signaling activity, and promoting resistance to Trastuzumab in HER2-positive breast cancer. High CMTM6 expression was associated not only with in vitro resistance phenotypes and more aggressive malignant behavior, but also with poor in vivo responses to Trastuzumab (alone or in combination with Pertuzumab), indicating that CMTM6 may serve as an important biomarker for predicting Trastuzumab efficacy and guiding intensified therapeutic strategies. [4]
In this study, treatment with Trastuzumab alone significantly inhibited the growth of SKBR3 tumors and CMTM6-silenced JIMT-1 tumors, but had no effect on control JIMT-1 tumors with high CMTM6 expression. The combination of Pertuzumab further enhanced the therapeutic effect of Trastuzumab in SKBR3 and CMTM6-silenced JIMT-1 tumors. Compared with control JIMT-1 tumors, CMTM6-silenced JIMT-1 tumors showed markedly reduced expression of CMTM6, HER2, and Ki67 proteins, along with increased expression of caspase-3. In addition, the relative expression levels of CMTM6, HER2, p-HER2, PI3K, AKT, MEK, and ERK were decreased in CMTM6-silenced JIMT-1 tumors compared with controls, and this effect was more pronounced after Trastuzumab treatment. These data indicate that CMTM6 promotes Trastuzumab resistance in breast cancer by stabilizing HER2 protein and enhancing downstream signaling.
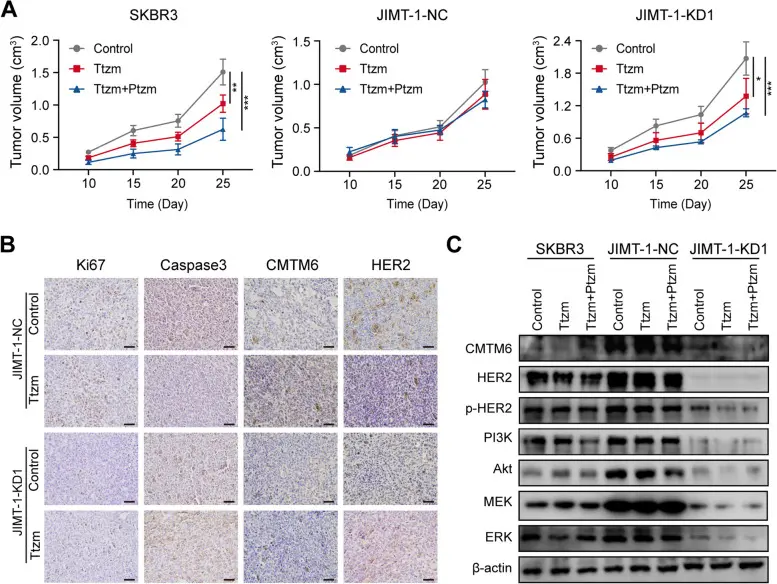
CMTM6 promotes the growth of Trastuzumab-resistant breast cancer by maintaining HER2 stability and downstream signaling in vivo [4]
Inhibition of DPAGT1 suppresses HER2 shedding and trastuzumab resistance in human breast cancer

Research Overview:
This study found that upregulation of dolichol-phosphate N-acetylglucosamine transferase (DPAGT1) sustains high levels of HER2 shedding, thereby leading to resistance to Trastuzumab. Following Trastuzumab treatment, membrane-associated DPAGT1 undergoes endocytosis via the caveolae pathway and is retrogradely transported to the endoplasmic reticulum (ER). In the ER, DPAGT1 promotes N-glycosylation of the sheddase ADAM10, ensuring its proper expression, maturation, and activation. N-glycosylation of ADAM10 at the N267 site protects it from ER-associated degradation, and this modification is critical for DPAGT1-mediated HER2 shedding and Trastuzumab resistance. Inhibition of DPAGT1 with tunicamycin, when combined with Trastuzumab, synergistically blocks HER2 signaling and reverses resistance. These findings reveal an important molecular mechanism underlying HER2 shedding and suggest that targeting DPAGT1 may represent a promising strategy to overcome Trastuzumab resistance in breast cancer. [5]
This study evaluated the therapeutic effect of combining tunicamycin with trastuzumab in HER2-positive breast cancer. Inhibition of DPAGT1 by tunicamycin significantly enhanced the sensitivity of SK-BR-3/DPAGT1 and BT-474/DPAGT1 cells to trastuzumab treatment, and the efficacy of the combination therapy was further confirmed in vivo using mouse models.
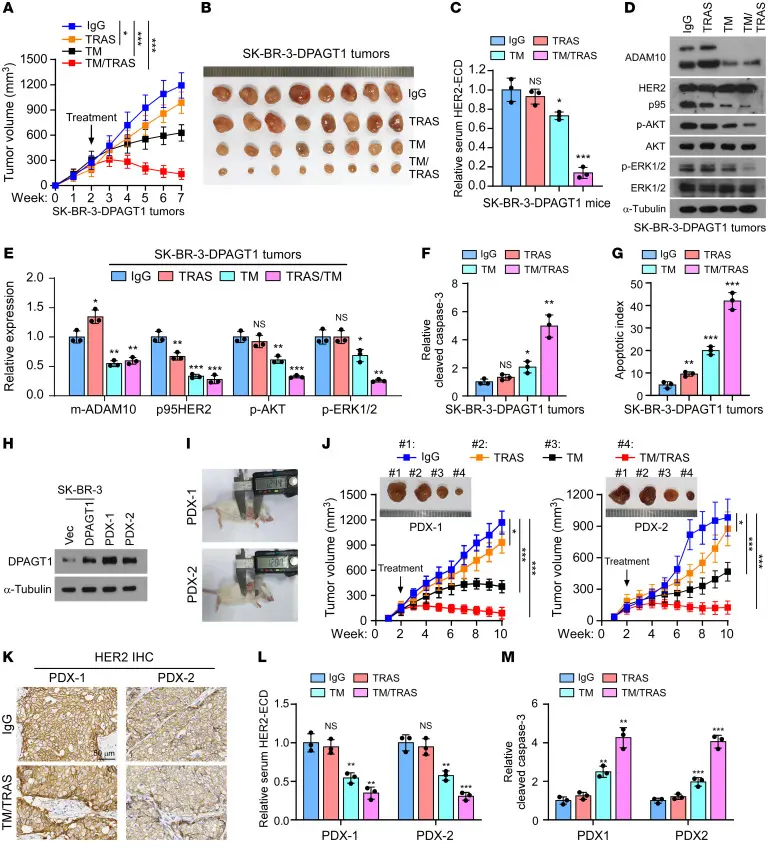
Inhibition of DPAGT1 can reverse the trastuzumab-resistant phenotype in breast cancer [5]
4. References
[1] Zhu K, Yang X, Tai H, et al. HER2-targeted therapies in cancer: a systematic review. Biomark Res. 2024 Feb 2;12(1):16. doi: 10.1186/s40364-024-00565-1. PMID: 38308374; PMCID: PMC10835834.
[2] Maadi H, Soheilifar MH, Choi WS, Moshtaghian A, Wang Z. Trastuzumab Mechanism of Action; 20 Years of Research to Unravel a Dilemma. Cancers (Basel). 2021 Jul 15;13(14):3540. doi: 10.3390/cancers13143540. PMID: 34298754; PMCID: PMC8303665.
[3] Panaampon J, Sungwan P, Fujikawa S, et al. Trastuzumab, a monoclonal anti-HER2 antibody modulates cytotoxicity against cholangiocarcinoma via multiple mechanisms. Int Immunopharmacol. 2024 Sep 10;138:112612. doi: 10.1016/j.intimp.2024.112612. Epub 2024 Jul 4. PMID: 38968862.
[4] Xing F, Gao H, Chen G, et al. CMTM6 overexpression confers trastuzumab resistance in HER2-positive breast cancer. Mol Cancer. 2023 Jan 10;22(1):6. doi: 10.1186/s12943-023-01716-y. Erratum in: Mol Cancer. 2024 Jan 5;23(1):3. doi: 10.1186/s12943-023-01918-4. Erratum in: Mol Cancer. 2025 Apr 14;24(1):115. doi: 10.1186/s12943-025-02317-7. PMID: 36627608; PMCID: PMC9830830.
[5] Yang M, Li Y, Kong L, et al. Inhibition of DPAGT1 suppresses HER2 shedding and trastuzumab resistance in human breast cancer. J Clin Invest. 2023 Jul 17;133(14):e164428. doi: 10.1172/JCI164428. PMID: 37463446; PMCID: PMC10348774.
Other Articles
Subscription to TargetMol News
An essential round-up of science news, opinion and analysis, delivered to your inbox every weekday.

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.