Your shopping cart is currently empty
Your shopping cart is currently empty
TargetMol—Disease Modeling—Lipopolysaccharides (Cat. No. T11855), Let the Immune Cells into Full Combat Readiness
1. Product Introduction
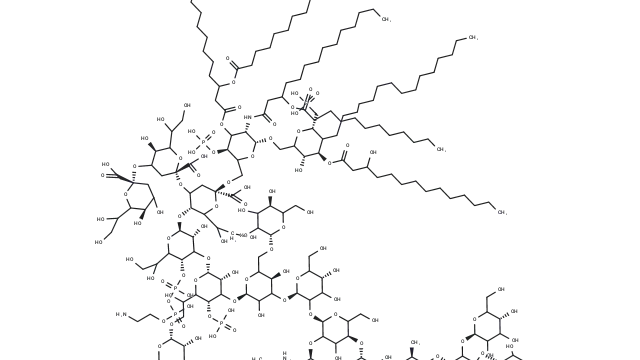
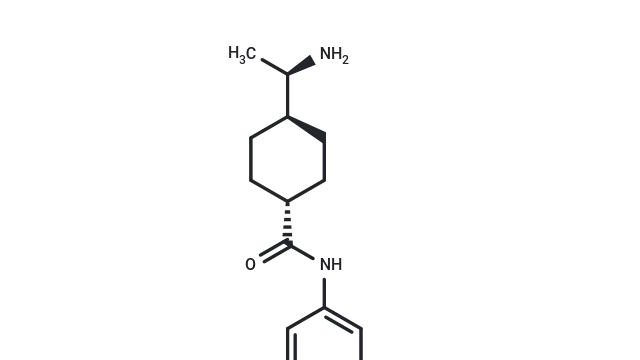
Lipopolysaccharides (Cat. No. T11855), also known as LPS. Lipopolysaccharides (LPS), derived from Escherichia coli O55: B5, is an important component of the outer membrane of Gram-negative bacteria. It is composed of lipid A, core oligosaccharides and O-specific polysaccharides. It has high immunogenicity. It can activate the TLR4 receptor of immune cells, induce the secretion of cell migration, resist the destruction of bile salts and lipid antibiotics. It is commonly used in the construction of inflammatory models, such as arthritis, chronic obstructive pulmonary disease (COPD), acute respiratory distress syndrome (ARDS), and digestive system disease models.
Molecular structure of Lipopolysaccharides
2. Background Introduction
Lipopolysaccharide (LPS) is an important component of the outer membrane of Gram-negative bacteria. It is widely found in bacteria such as Escherichia coli O55: B5. It is composed of lipid A, core oligosaccharides and O-specific polysaccharides. Lipid A is the main structural basis of its immune activity. LPS has a highly conserved molecular pattern and can act as a typical pathogen-associated molecular pattern (PAMP) that is recognized by the host innate immune system and rapidly induces an inflammatory response. In the process of immune recognition, LPS is first extracted and transported to CD14 by LPS binding protein (LBP) in serum, and then delivered to TLR4 / MD-2 receptor complex, triggering downstream signaling cascades. As a key pattern recognition receptor (PRR), TLR4 is a core target for mediating LPS-induced inflammatory response. Its activation can drive the establishment of a variety of infection and inflammation-related disease models, including arthritis, COPD and ARDS. [1]
LPS-mediated immune activation mainly depends on the stepwise amplification of TLR4 signal transduction pathway. Extracellularly, LPS is transported by LBP, binds to CD14, and is delivered to the TLR4-MD-2 complex to induce receptor dimerization and initiate intracellular signal transduction. Subsequently, TLR4 activates the downstream signaling network through two classical pathways: MyD88-dependent pathway and MyD88-independent pathway. MyD88-dependent pathway mainly activates IRAKs and TRAF6, and further initiates NF-κB and AP-1 transcription factors, thereby inducing the expression of pro-inflammatory factors such as TNF-α and IL-6. The TRIF-dependent pathway activates IRF3 and promotes type I interferon production. The synergistic effect of the two pathways leads to macrophage M1 polarization and a strong inflammatory cascade, which is the key mechanism basis of LPS-induced acute inflammatory injury and various inflammatory diseases (such as ARDS, sepsis). [2]
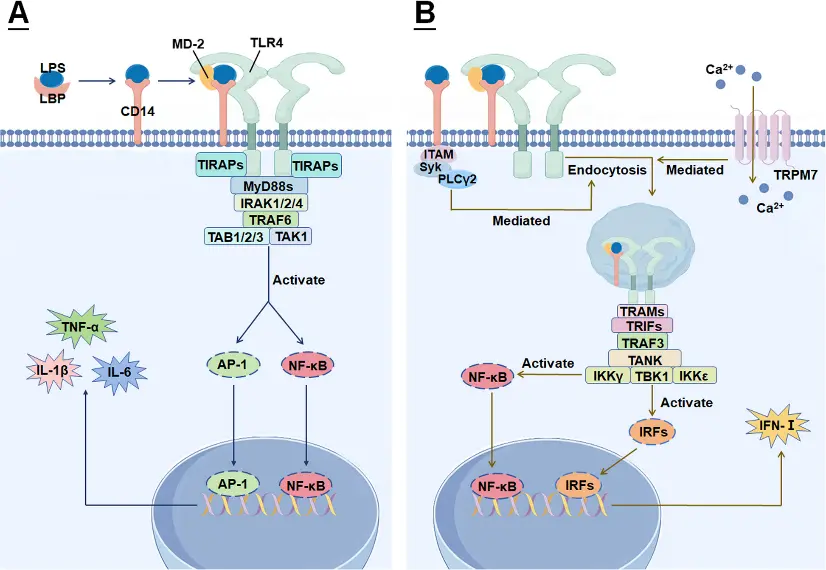
LPS-TLR4 mediated signal transduction pathway [2]
3. Application References
Hyperglycemia enhances brain susceptibility to lipopolysaccharide-induced neuroinflammation via astrocyte reprogramming

Research Overview:
This study systematically explored how hyperglycemia amplifies Lipopolysaccharide-induced central nervous system inflammation in vitro and in vivo. The authors combined hyperglycemia treatment with Lipopolysaccharide stimulation in a rat model and found that high glucose environment significantly enhanced the sensitivity of brain tissue to inflammatory stimulation, manifested as stronger release of inflammatory factors, glial cell activation, and nerve tissue damage. In terms of mechanism, the study focused on revealing that astrocytes undergo metabolic and phenotypic ' reprogramming ' under high glucose conditions, transforming from steady-state supporting cells to pro-inflammatory reaction-amplifying cells, thereby significantly amplifying TLR4-mediated inflammatory cascades under the action of Lipopolysaccharide. In vitro cultured astrocytes experiment further verified that high glucose can enhance its responsiveness to Lipopolysaccharide and promote inflammation-related signaling pathways and cytokine expression. [3]
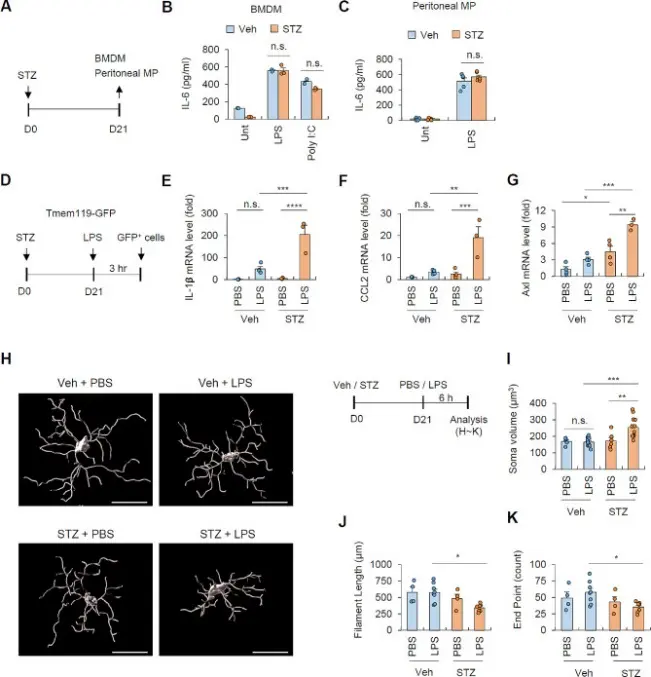
Hyperglycemia increases the sensitivity of microglia to systemic LPS administration [3]
Circadian Gene BMAL1 Regulation of Cellular Senescence in Thyroid Aging

Research Overview:
This study focuses on the role of circadian rhythm core gene BMAL1 in thyroid aging, and reveals its regulatory mechanism on cell aging process through in vivo and in vitro experiments. The expression of BMAL1 in thyroid tissues at different ages was compared. It was found that the expression of BMAL1 decreased significantly with age and was closely related to the increase of cell senescence markers. Further, in the thyroid epithelial cell model, by interfering with the expression of BMAL1, it was confirmed that it could regulate cell cycle arrest, oxidative stress level and the expression of inflammation-related factors, thus affecting the cell senescence phenotype. Mechanism studies have shown that BMAL1 maintains the stability of thyroid cell function and delays the aging process by regulating redox homeostasis and related signaling pathways. This study shows that circadian rhythm disorders accelerate thyroid cell aging and aggravate thyroid function decline, which provides a new theoretical basis for understanding the mechanism of thyroid aging and maintaining thyroid function stability. [4]
In this study, Lipopolysaccharides (Cat. No. T11855) were mainly used as a cell stimulator to induce cell inflammatory response and cell senescence-related processes. This experiment helped to distinguish that the decrease of BMAL1 was an upstream event of aging, not caused by inflammation activated by Lipopolysaccharides, and supported the independent role of BMAL1 in the regulation of cell aging.
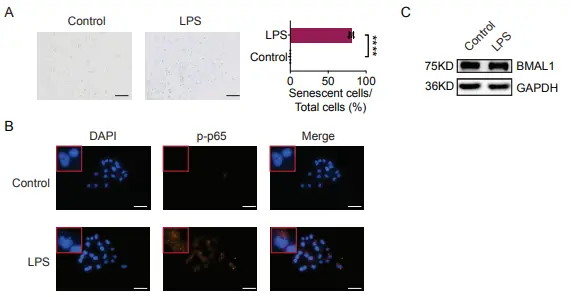
LPS induced HTori-3.1 cell senescence and NF-κB activation, but did not inhibit BMAL1 expression [4]
4. References
[1] Park BS, Lee JO. Recognition of lipopolysaccharide pattern by TLR4 complexes. Exp Mol Med. 2013 Dec 6;45(12):e66. doi: 10.1038/emm.2013.97. PMID: 24310172; PMCID: PMC3880462.
[2] Luo R, Yao Y, Chen Z, Sun X. An examination of the LPS-TLR4 immune response through the analysis of molecular structures and protein-protein interactions. Cell Commun Signal. 2025 Mar 18;23(1):142. doi: 10.1186/s12964-025-02149-4. PMID: 40102851; PMCID: PMC11921546.
[3] Lee KS, Yoon SH, Hwang I, Ma JH, Yang E, Kim RH, Kim E, Yu JW. Hyperglycemia enhances brain susceptibility to lipopolysaccharide-induced neuroinflammation via astrocyte reprogramming. J Neuroinflammation. 2024 May 27;21(1):137. doi: 10.1186/s12974-024-03136-1. PMID: 38802820; PMCID: PMC11131277.
[4] Zong D, Sun B, Ye Q, Cao H, Guan H. Circadian Gene BMAL1 Regulation of Cellular Senescence in Thyroid Aging. Aging Cell. 2025 Aug;24(8):e70119. doi: 10.1111/acel.70119. Epub 2025 May 28. PMID: 40434135; PMCID: PMC12341809.
Other Articles
Subscription to TargetMol News
An essential round-up of science news, opinion and analysis, delivered to your inbox every weekday.

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.