Your shopping cart is currently empty
Your shopping cart is currently empty
TargetMol—Signaling Pathway—Doxorubicin hydrochloride (Cat. No. T1020, CAS. 25316-40-9), DNA-embedded Cell Killer
1. Product Introduction
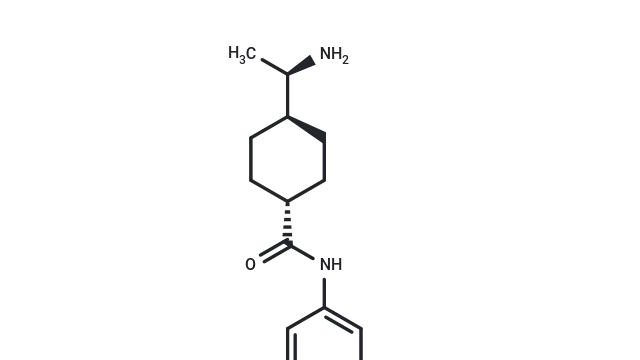
Doxorubicin hydrochloride (Cat. No. T1020, Cas. 25316-40-9), also known as NSC 123127, Hydroxydaunorubicin hydrochloride, DOX hydrochloride, Adriamycin HCl. Doxorubicin hydrochloride is an anthracycline antibiotic with cytotoxic and antitumor activity. It is an effective inhibitor of human DNA topoisomerase II with IC50 values of 0.8 μM and 2.67 μM, respectively. It can induce apoptosis and autophagy. In animal experiments, Doxorubicin hydrochloride is often used to induce acute renal failure, chronic kidney injury and heart failure models.
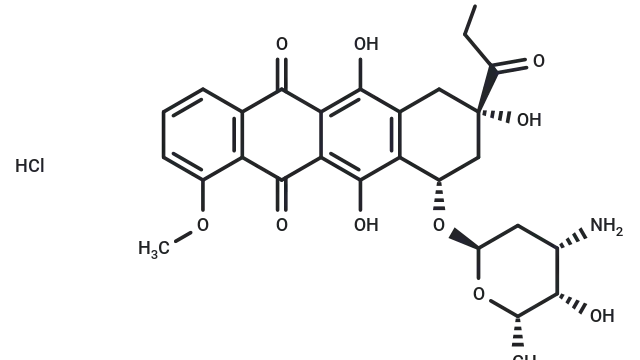
Molecular structure of Doxorubicin hydrochloride
2. Background Introduction
DNA topoisomerase (Topo) is a key enzyme that regulates the supercoiled state and topological structure of DNA, and plays a central role in DNA replication, transcription, recombination and repair. Topo I relieves the torsional tension through reversible single-strand cleavage, while Topo II solves the problem of DNA entanglement and chromosome separation through reversible double-strand breaks. Due to the high proliferation of tumor cells, the dependence on topoisomerase activity is significantly enhanced, so the enzyme has become an important anti-tumor therapeutic target. By stabilizing the ' Topo-DNA cleavage complex ', topoisomerase inhibitors block the reconnection after DNA cleavage, causing persistent DNA damage and activating the cell death pathway. [1]
Doxorubicin hydrochloride, as a classical anthracycline antitumor drug, has a multi-channel synergistic cytotoxicity mechanism. Its anthracycline planar structure can be embedded between DNA base pairs to interfere with the double helix conformation, while stabilizing the Topo II-DNA cleavage complex, resulting in the accumulation of double-stranded DNA cleavage and inhibiting DNA replication and transcription. In addition, the drug can induce apoptosis through mitochondrial damage and reactive oxygen species (ROS) generation, and regulate the autophagy process in a variety of models. Furthermore, studies have shown that Doxorubicin can interfere with cellular energy metabolism signals, affect the phosphorylation status of AMPK and its downstream ACC, aggravate energy stress and promote programmed cell death. These mechanisms together constitute the basis of its broad-spectrum and potent anti-tumor activity. [2]
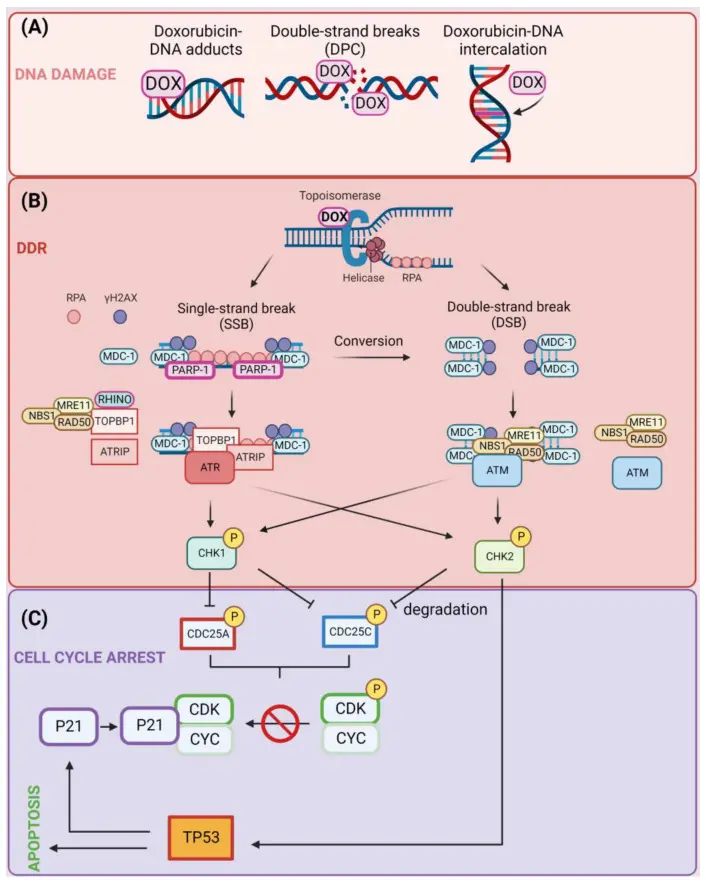
Induction of DNA damage by Doxorubicin (DOX) [2]
3. Application References
Doxorubicin downregulates autophagy to promote apoptosis-induced dilated cardiomyopathy via regulating the AMPK/mTOR pathway

Research Overview:
In this study, the key molecular mechanism of Doxorubicin-induced dilated cardiomyopathy was elucidated by in vivo mouse model and in vitro cardiomyocyte experiment system. It was found that Doxorubicin treatment significantly increased the level of ROS in cardiomyocytes and led to mitochondrial dysfunction, while inhibiting the activation of energy sensing pathway AMPK and its downstream mTOR signaling, thereby inhibiting the protective autophagy process. After autophagy is blocked, damaged mitochondria and oxidative stress cannot be effectively removed, which ultimately aggravates caspase-dependent apoptosis and leads to myocardial structural and functional damage. Further pharmacological or genetic restoration of AMPK activity or enhancement of autophagy can significantly reduce Doxorubicin-induced cardiomyocyte apoptosis and cardiac dysfunction. This study revealed the core regulatory role of the ' AMPK / mTOR-autophagy-apoptosis ' axis in Doxorubicin cardiotoxicity and provided a potential intervention target for alleviating its cardiotoxicity. [3]
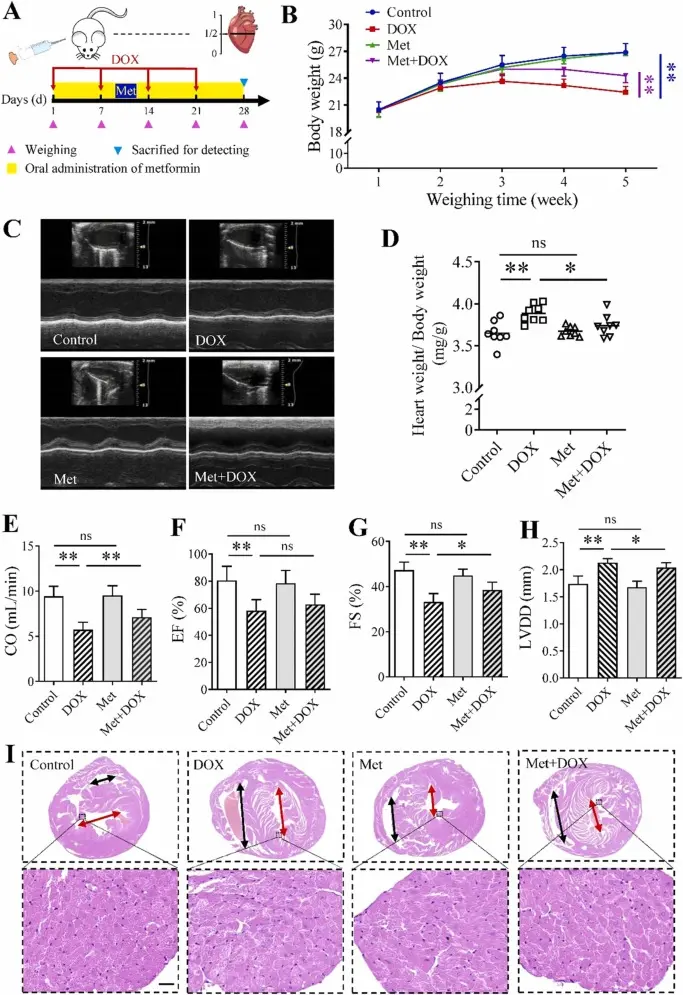
DOX inhibits AMPK activity and induces dilated cardiomyopathy by down-regulating autophagy to promote apoptosis [3]
USP13 dictates Ran turnover and vulnerability to ferroptosis in diffuse large B cell lymphoma (DLBCL)

Research Overview:
This study revealed the key mechanism by which the deubiquitinating enzyme USP13 affects the sensitivity of cells to ferroptosis by regulating the protein stability of the small GTPase Ran in a diffuse large B-cell lymphoma (DLBCL) model. The authors found that USP13 can remove the ubiquitination modification of Ran and maintain its protein homeostasis; when USP13 was inhibited or knocked down, Ran protein accelerated degradation, resulting in impaired nuclear-cytoplasmic transport homeostasis and triggering intracellular iron homeostasis disorders and lipid peroxidation accumulation, thereby significantly enhancing the sensitivity of DLBCL cells to ferroptosis inducers. Further in vitro and in vivo experiments showed that the USP13-Ran axis is an important molecular pathway that regulates the anti-iron death ability of DLBCL cells, and targeting USP13 can be used as a potential new way to improve the responsiveness of tumors to ferroptosis treatment strategies. [4]
In this study, Doxorubicin (Cat. No. T1020) was used as a conventional chemotherapeutic drug control and combined treatment tool for DLBCL cells. Doxorubicin was used to induce DNA damage and cellular stress to evaluate the changes in the response of DLBCL cells to chemotherapy in the context of USP13 inhibition or knockdown.
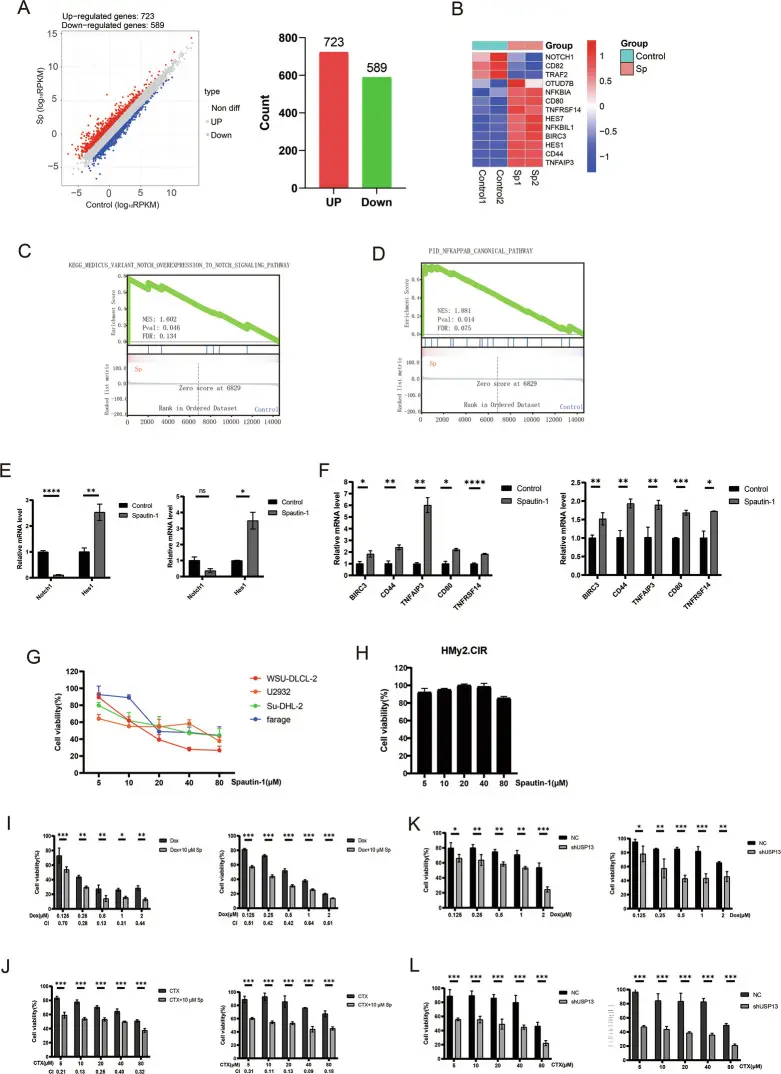
Spautin-1 inhibits the survival of DLBCL cells and has a synergistic effect with Doxorubicin (Dox) or Cyclophosphamide (CTX) [4]
4. References
[1] Pommier Y. Topoisomerase I inhibitors: camptothecins and beyond. Nat Rev Cancer. 2006 Oct;6(10):789-802. doi: 10.1038/nrc1977. PMID: 16990856.
[2] Kciuk M, Gielecińska A, Mujwar S, Kołat D, Kałuzińska-Kołat Ż, Celik I, Kontek R. Doxorubicin-An Agent with Multiple Mechanisms of Anticancer Activity. Cells. 2023 Feb 19;12(4):659. doi: 10.3390/cells12040659. PMID: 36831326; PMCID: PMC9954613.
[3] Zhang S, Wei X, Zhang H, Wu Y, Jing J, Huang R, Zhou T, Hu J, Wu Y, Li Y, You Z. Doxorubicin downregulates autophagy to promote apoptosis-induced dilated cardiomyopathy via regulating the AMPK/mTOR pathway. Biomed Pharmacother. 2023 Jun;162:114691. doi: 10.1016/j.biopha.2023.114691. Epub 2023 Apr 14. PMID: 37060659.
[4] Qiao X, Yang X, Diao Y, Li Q, Wang X, Li C, Yang Z, Chng WJ, Li B. USP13 dictates Ran turnover and vulnerability to ferroptosis in diffuse large B cell lymphoma (DLBCL). Cell Death Dis. 2025 Nov 28;16(1):870. doi: 10.1038/s41419-025-08207-6. PMID: 41315297; PMCID: PMC12663190.
Other Articles
Subscription to TargetMol News
An essential round-up of science news, opinion and analysis, delivered to your inbox every weekday.

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.