Your shopping cart is currently empty
Your shopping cart is currently empty
TargetMol—Ubiquitylation—MG-132 (Cat. No. T2154, CAS 133407-82-6), Multi-Pathway Regulation of Apoptosis
1. Product Introduction
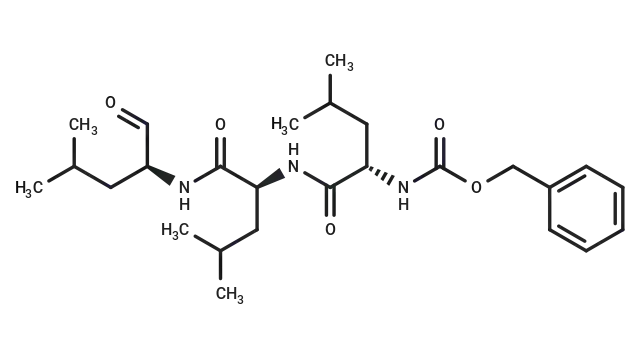
MG-132 (Cat. No. T2154, CAS. 133407-82-6), also known as Z-LLL-al,Z-Leu-Leu-Leu-CHO,MG132. MG-132 is a 26S proteasome inhibitor (IC50 = 100 nM) with cell permeability and reversibility. MG-132 can be used as an autophagy activator to induce apoptosis.
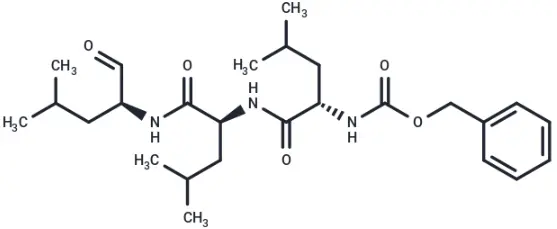
Molecular structure of MG-132
2. Background Introduction
The 26S proteasome is the most important protein degradation system in cells, which is responsible for the selective degradation of ubiquitinated proteins and plays a central role in maintaining protein homeostasis, regulating cell cycle, signal transduction and stress response. The ubiquitin-proteasome system (UPS) not only participates in the renewal of short-lived proteins, but also degrades a large number of long-lived proteins, which is one of the key pathways for intracellular protein quality control. Its functional imbalance is closely related to various pathological processes such as tumorigenesis, neurodegenerative diseases and inflammatory reactions, so it is considered to be an important drug intervention target. [1]
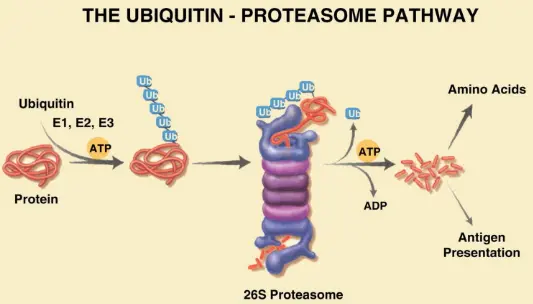
Schematic diagram of ubiquitin-proteasome pathway [1]
MG-132 is a peptide aldehyde 26S proteasome inhibitor with cell permeability and reversibility. It mainly inhibits its proteolytic activity by reversibly binding to the 20S proteasome β5 subunit, leading to the accumulation of ubiquitinated proteins in cells and inducing protein homeostasis imbalance. This process can trigger endoplasmic reticulum stress and oxidative stress, and then activate multiple signaling pathways (such as JNK, MAPK, etc.) to promote apoptosis. At the same time, proteasome inhibition can also induce autophagy as a compensation mechanism, which promotes the formation of autophagosomes by up-regulating autophagy-related proteins such as Beclin-1 and LC3. Under certain conditions, autophagy can not only be used as a protective mechanism to alleviate protein toxicity, but also coordinate with apoptosis to regulate cell fate. [2]
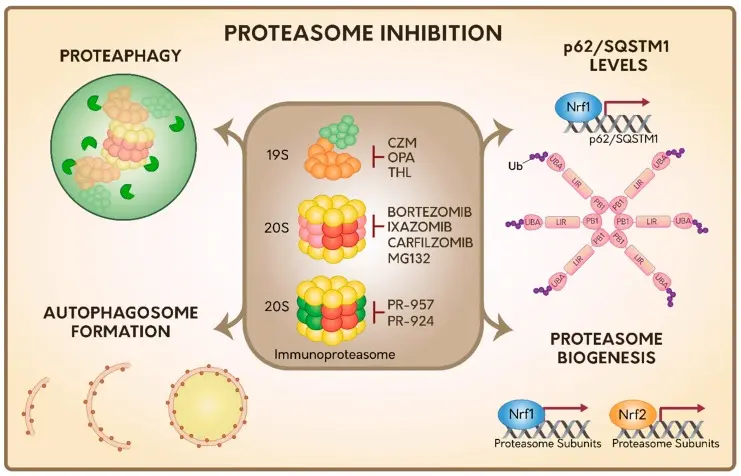
Summary of cell responses during proteasome inhibition [2]
3. Application References
MG-132 activates sodium palmitate-induced autophagy in human vascular smooth muscle cells and inhibits senescence via the PI3K/AKT/mTOR axis

Research Overview:
This study focuses on the regulatory role of proteasome inhibitor MG-132 in lipid-induced cell damage, and uses sodium palmitate to induce stress and senescence phenotypes in human vascular smooth muscle cells (VSMCs). The results showed that MG-132 significantly activated the autophagy process by inhibiting proteasome activity, which was manifested by increased LC3-II level and up-regulation of autophagy-related protein expression. Mechanistically, it mainly relieves the negative regulation of autophagy by inhibiting the PI3K/AKT/mTOR signaling pathway. At the same time, MG-132 treatment can effectively reduce cell senescence-related markers (such as β-gal activity) and improve cell functional status. Further studies suggest that autophagy plays a key mediating role in this process, and inhibition of autophagy will weaken the anti-aging effect of MG-132. This study reveals the molecular mechanism of MG-132 by 'inhibiting PI3K/AKT/mTOR-activating autophagy-delaying cell senescence', and provides a theoretical basis for its potential application in metabolic-related vascular diseases. [3]
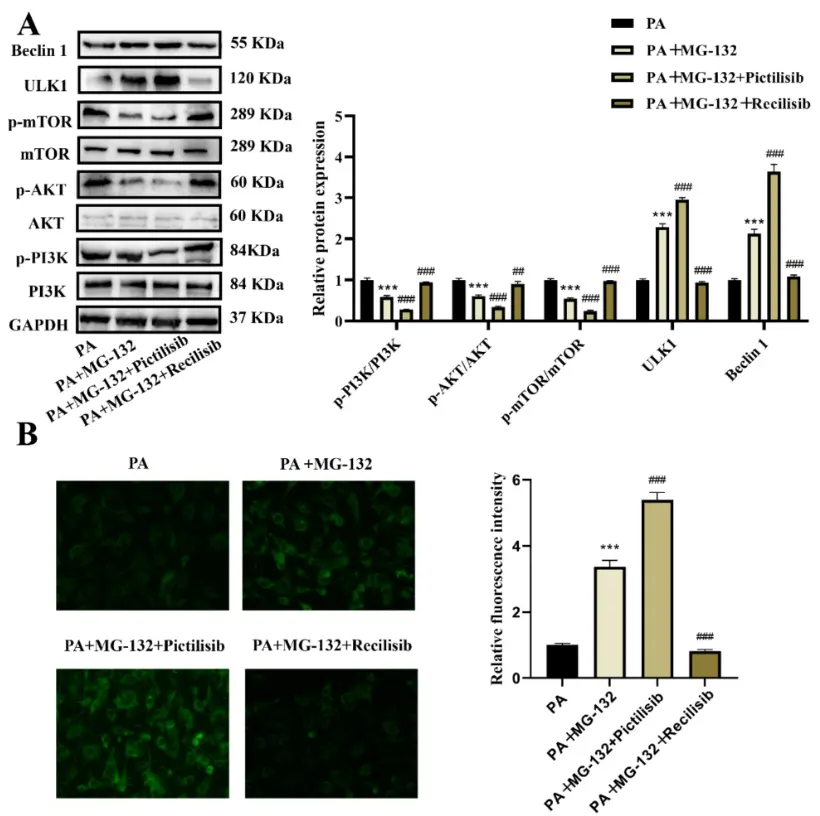
MG-132 promotes autophagy of VSMCs induced by sodium palmitate by inhibiting the PI3K/AKT/mTOR pathway. [3]
Proteasome inhibitors induce apoptosis by superoxide anion generation via NADPH oxidase 5 in human neuroblastoma SH-SY5Y cells

Research Overview:
In this study, human neuroblastoma cell line SH-SY5Y was used as a model to investigate the molecular mechanism of apoptosis induced by proteasome inhibitors (such as MG-132). It has been found that proteasome inhibition can significantly promote the production of superoxide anion in cells and further trigger the process of apoptosis. By inhibiting NOX5 or scavenging reactive oxygen species (ROS), the level of oxidative stress and apoptosis induced by proteasome inhibitors can be significantly reduced, indicating that NOX5-mediated ROS generation plays a key role in this process. It was also found that MG-132 treatment increased the expression of NOX5 (a member of the NOX family), while siRNA-mediated NOX5 and BAPTA-AM silencing of NOX5 by chelating calcium inhibited MG-132-induced apoptosis and reactive oxygen species production in SH-SY5Y cells. [4]
Through Western blot, RT-PCR and RNA interference (siRNA), it was proved that the proteasome inhibitor MG-132 could up-regulate the expression of transcription factor GATA2 in SH-SY5Y cells, and then drive the mRNA transcription of NOX5. The experimental results emphasize that GATA2 is a key intermediate molecule that mediates MG-132-induced NOX5 expression. After knocking down GATA2, the effect of MG-132-induced NOX5 increase is significantly weakened.
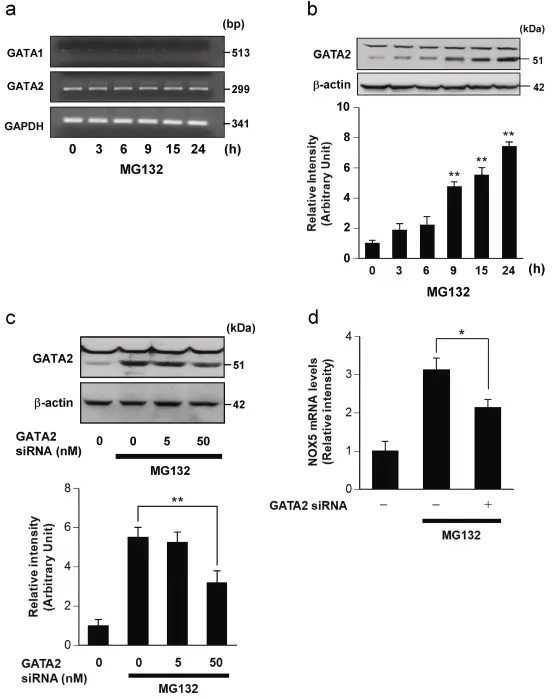
Proteasome inhibitor ( MG-132 ) induced NOX5 mRNA expression by increasing GATA2 expression in SH-SY5Y cells. [4]
4. References
[1] Kisselev AF, Goldberg AL. Proteasome inhibitors: from research tools to drug candidates. Chem Biol. 2001 Aug;8(8):739-58. doi: 10.1016/s1074-5521(01)00056-4. PMID: 11514224.
[2] Albornoz N, Bustamante H, Soza A, et al. Cellular Responses to Proteasome Inhibition: Molecular Mechanisms and Beyond. Int J Mol Sci. 2019 Jul 10;20(14):3379. doi: 10.3390/ijms20143379. PMID: 31295808; PMCID: PMC6678303.
[3] Shu Z, Li X, Zhang W, et al. MG-132 activates sodium palmitate-induced autophagy in human vascular smooth muscle cells and inhibits senescence via the PI3K/AKT/mTOR axis. Lipids Health Dis. 2024 Sep 4;23(1):282. doi: 10.1186/s12944-024-02268-w. PMID: 39232759; PMCID: PMC11373134.
[4] Yamamuro-Tanabe A, Oshima Y, Iyama T, et al. Proteasome inhibitors induce apoptosis by superoxide anion generation via NADPH oxidase 5 in human neuroblastoma SH-SY5Y cells. J Pharmacol Sci. 2024 Jun;155(2):52-62. doi: 10.1016/j.jphs.2024.03.002. Epub 2024 Mar 27. PMID: 38677786.
Other Articles
Subscription to TargetMol News
An essential round-up of science news, opinion and analysis, delivered to your inbox every weekday.

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.