Your shopping cart is currently empty
Your shopping cart is currently empty
TargetMol—Signaling Pathway—Dexamethasone (Cat. No. T1076, CAS. 50-02-2), Precisely Strike Inflammatory Cytokines
1. Product Introduction
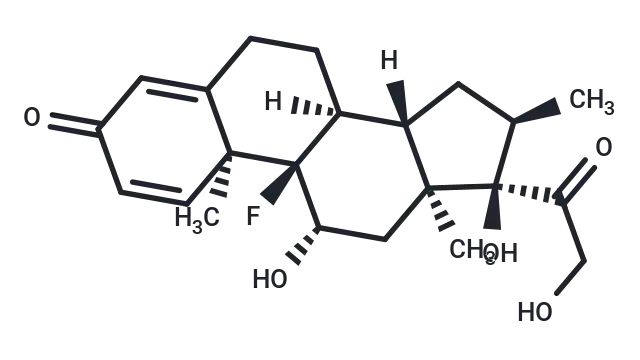
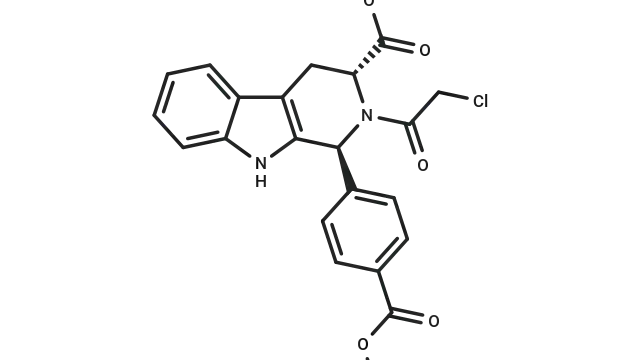
Dexamethasone (Cat. No. T1076, CAS. 50-02-2), also known as Prednisolone F, NSC 34521, MK 125, Hexadecadrol. Dexamethasone is a glucocorticoid receptor agonist. It has anti-inflammatory, immunosuppressive and apoptosis-inducing activities. It can inhibit the production of inflammatory miRNA-155 exosomes in macrophages, significantly reduce the expression of inflammatory factors in neutrophils and monocytes, inhibit LPS-induced macrophage inflammatory response and induce autophagy. It excess use causes depression, muscle atrophy and hypertension animal models, and has potential in COVID-19 research.
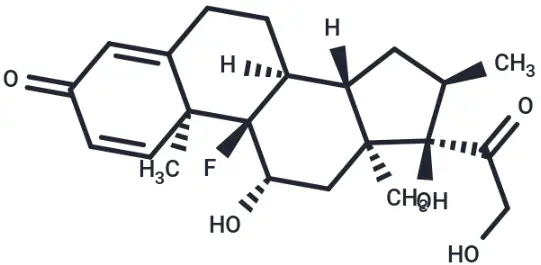
Molecular structure of Dexamethasone
2. Background Introduction
Glucocorticoid receptor (GR) is a ligand-activated transcriptional regulatory belonging to the nuclear receptor superfamily, which is widely expressed in a variety of immune cells including macrophages, monocytes and neutrophils. When glucocorticoids (e.g., dexamethasone) enter cells, they bind to glucocorticoid receptors in the cytoplasm, causing the glucocorticoid receptors to dissociate the accompanying molecular chaperone proteins (e.g., Hsp90) and translocate into the nucleus. Glucocorticoid receptors activated in the nucleus can directly bind to the glucocorticoid response element (GRE) on DNA to initiate or inhibit the transcription of target genes, thereby regulating multiple signaling pathways including inflammatory factors, immune regulation-related molecules, and apoptosis-related genes. These actions make glucocorticoid receptors a central regulatory target for controlling the inflammatory response, immune response, and cell survival / death balance. Glucocorticoid receptor activation can not only up-regulate the expression of anti-inflammatory proteins, but also inhibit the activity of pro-inflammatory transcription factors such as NF-κB, thereby extensively inhibiting the transcription of inflammatory genes. At the same time, studies in macrophages have shown that glucocorticoids can significantly reduce the release of exosomes containing inflammatory miRNA-155 induced by lipopolysaccharide (LPS). This mechanism helps to reduce macrophage-mediated inflammatory factor expression and intercellular inflammatory signal transmission. [1]
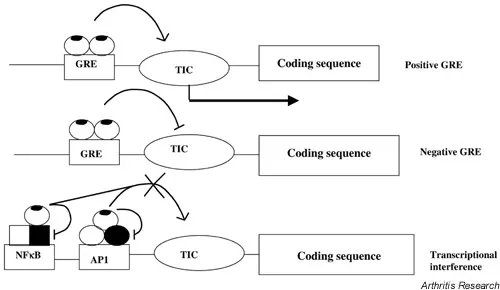
Glucocorticoid transcriptional regulation mechanism [2]
Dexamethasone is a synthetic glucocorticoid receptor agonist with strong anti-inflammatory, immunosuppressive and pro-apoptotic activities. The classical mechanism is to regulate the expression of target genes after binding to glucocorticoid receptors, thereby inhibiting inflammatory response and immune cell function. Activated glucocorticoid receptor complex can up-regulate anti-inflammatory proteins (such as Annexin-1, MAPK phosphatase) by transcriptional activation and block the expression and activity of pro-inflammatory factors (such as TNF-α, IL-6) and inflammation-related transcription factor NF-κB by transcriptional inhibition to achieve a wide range of anti-inflammatory and immunosuppressive effects. In the macrophage model, Dexamethasone treatment significantly reduced LPS-induced secretion of inflammatory exosomes and down-regulated the expression of miRNA-155, which is closely related to its anti-inflammatory effect, because miRNA-155 is an inflammatory promoting factor and can enhance the production of inflammatory mediators. In addition, Dexamethasone can induce autophagy and apoptosis processes, such as promoting autophagy and mitochondrial-related mechanisms through the glucocorticoid receptor pathway in muscle cells, and promoting apoptosis in certain immune cells to achieve immunosuppressive effects. Therefore, in addition to the classic glucocorticoid receptor-mediated gene regulation, Dexamethasone also exerts a comprehensive immunomodulatory effect by regulating intracellular autophagy, promoting apoptosis and inhibiting inflammatory signal transduction. [3, 4]
3. Application References
Dexamethasone impairs the expression of antimicrobial mediators in lipopolysaccharide-activated primary macrophages by inhibiting both expression and function of interferon β

Research Overview:
In this study, LPS-activated primary mouse macrophages were used as an in vitro inflammatory model to systematically explore the effect of Dexamethasone on the antibacterial defense function of macrophages and its molecular mechanism. Through transcriptome sequencing, protein detection and functional experiments, it was found that Dexamethasone not only inhibited the expression of inflammatory factors through the classical anti-inflammatory pathway, but also significantly inhibited the expression of LPS-induced type I interferon IFNβ and its signal transduction function. Since IFNβ is a key upstream signal that drives the expression of a variety of antibacterial effectors (such as chemokines, antibacterial proteins, and inflammatory regulators), Dexamethasone 's inhibition of the IFNβ transcription program further impairs the expression of a wide range of downstream antibacterial mediators. Studies have revealed that the important mechanism by which dexamethasone produces immunosuppressive effects in macrophages is not simply ' anti-inflammatory ', but by blocking the IFNβ-dependent antibacterial gene network, thereby weakening the innate immune defense ability of macrophages. This finding provides direct experimental evidence for understanding the molecular basis of glucocorticoids in increasing the risk of infection and immunosuppression. [5]
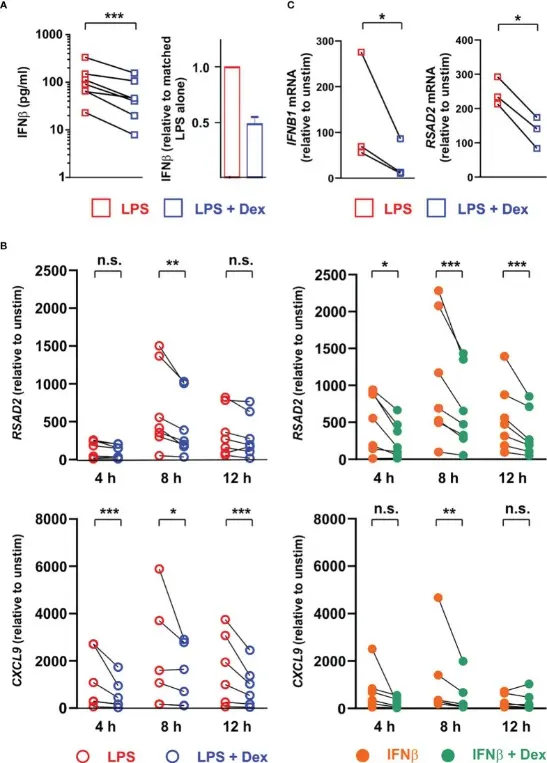
Dexamethasone regulates IFNβ‑dependent signaling in primary mouse macrophages[5]
GW8510 alleviates muscle atrophy and skeletal muscle dysfunction in mice through AMPK/PGC1α signaling

Research Overview:
This study systematically evaluated the improvement of GW8510 (a CDK2 inhibitor) on muscle atrophy and skeletal muscle dysfunction in a mouse model and its molecular mechanism. By establishing a denervated and glucocorticoid-induced muscle atrophy model, it was found that GW8510 treatment significantly improved skeletal muscle mass, muscle fiber cross-sectional area, and muscle strength performance. Mechanistic studies have shown that GW8510 can effectively activate the AMPK / PGC1α signaling pathway, enhance mitochondrial biogenesis and oxidative metabolism, promote energy metabolism reprogramming, and inhibit the ubiquitin-proteasome system associated with muscle protein degradation and excessive activation of autophagy. Further experiments showed that GW8510 reversed the process of muscle atrophy by improving mitochondrial function and metabolic homeostasis, reducing oxidative stress and inflammatory response. This study reveals a new mechanism by which GW8510 protects skeletal muscle structure and function by activating AMPK / PGC1α, and provides a potential pharmacological strategy for the intervention of muscle atrophy and related metabolic myopathy. [6]
In this study, Dexamethasone (Cat. No. T1076) was used as a chemical inducer to establish a muscle atrophy model. Skeletal muscle atrophy was induced in mice by continuous intraperitoneal injection of dexamethasone (10 mg / kg) to evaluate the protective effect of GW8510 on muscle atrophy and to study the potential mechanism. Using this dexamethasone-induced model, the study showed that GW8510 could improve muscle volume, muscle fiber cross-sectional area and muscle function, and reverse dexamethasone-induced atrophy-related gene expression and mitochondrial function damage, thereby verifying the potential efficacy of GW8510 in muscle atrophy intervention.
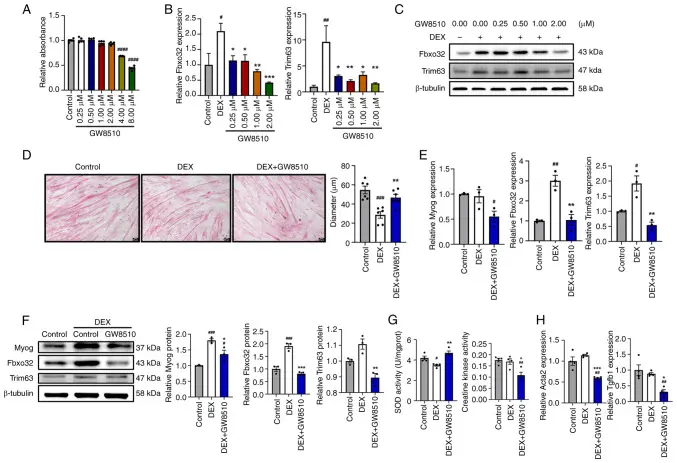
GW8510 ameliorated Dexamethasone-induced C2C12 myotube atrophy in vitro [6]
4. References
[1] Chen Y, Zhang M, Zheng Y. Glucocorticoids inhibit production of exosomes containing inflammatory microRNA-155 in lipopolysaccharide-induced macrophage inflammatory responses. Int J Clin Exp Pathol. 2018 Jul 1;11(7):3391-3397. PMID: 31949716; PMCID: PMC6962844.
[2] Saklatvala J. Glucocorticoids: do we know how they work? Arthritis Res. 2002;4(3):146-50. doi: 10.1186/ar398. Epub 2002 Jan 21. PMID: 12010562; PMCID: PMC128923.
[3] Molitoris JK, McColl KS, Swerdlow S, Matsuyama M, Lam M, Finkel TH, Matsuyama S, Distelhorst CW. Glucocorticoid elevation of dexamethasone-induced gene 2 (Dig2/RTP801/REDD1) protein mediates autophagy in lymphocytes. J Biol Chem. 2011 Aug 26;286(34):30181-9. doi: 10.1074/jbc.M111.245423. Epub 2011 Jul 6. Erratum in: J Biol Chem. 2011 Nov 11;286(45):39673. PMID: 21733849; PMCID: PMC3191057.
[4] Coutinho AE, Chapman KE. The anti-inflammatory and immunosuppressive effects of glucocorticoids, recent developments and mechanistic insights. Mol Cell Endocrinol. 2011 Mar 15;335(1):2-13. doi: 10.1016/j.mce.2010.04.005. Epub 2010 Apr 14. PMID: 20398732; PMCID: PMC3047790.
[5] O'Neil JD, Bolimowska OO, Clayton SA, Tang T, Daley KK, Lara-Reyna S, Warner J, Martin CS, Mahida RY, Hardy RS, Arthur JSC, Clark AR. Dexamethasone impairs the expression of antimicrobial mediators in lipopolysaccharide-activated primary macrophages by inhibiting both expression and function of interferon β. Front Immunol. 2023 Oct 24;14:1190261. doi: 10.3389/fimmu.2023.1190261. PMID: 37942320; PMCID: PMC10628473.
[6] Chen Y, Liu Z, Liu C, Yang D, Xiao M, Li Z, Xie Z. GW8510 alleviates muscle atrophy and skeletal muscle dysfunction in mice through AMPK/PGC1α signaling. Int J Mol Med. 2025 Sep;56(3):128. doi: 10.3892/ijmm.2025.5569. Epub 2025 Jun 27. PMID: 40576133; PMCID: PMC12236748.
Other Articles
Subscription to TargetMol News
An essential round-up of science news, opinion and analysis, delivered to your inbox every weekday.

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.