Your shopping cart is currently empty
Your shopping cart is currently empty
TargetMol—Natural Product—Rapamycin (Cat. No. T1537, CAS. 53123-88-9), a Classical Molecular Tool for Activating Autophagy and Metabolic Reprogramming
1. Product Introduction
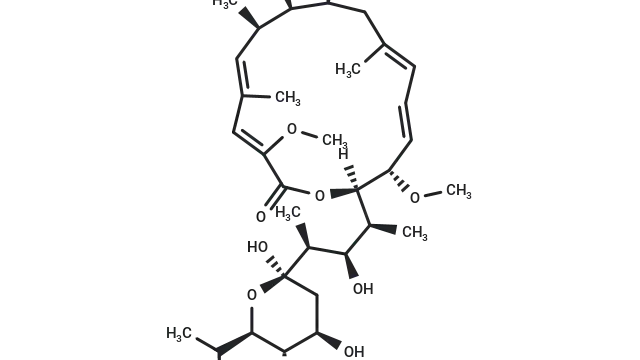
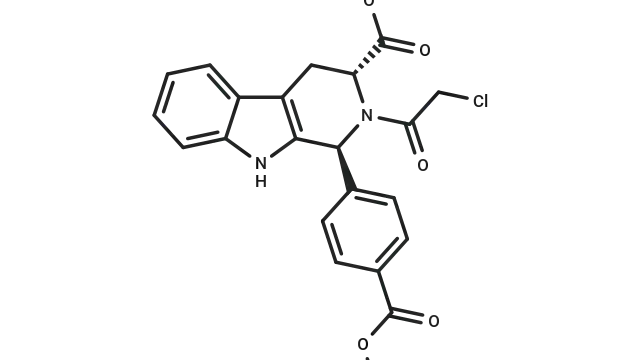
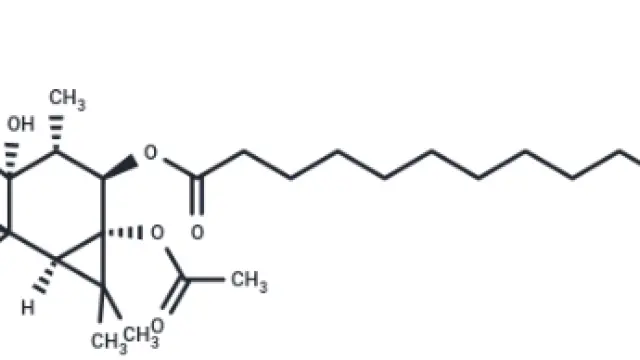
Rapamycin (Cat. No. T1537, CAS. 53123-88-9), also known as Sirolimus, NSC-2260804, AY 22989. Rapamycin (AY 22989), a natural product of macrolides, is a mTOR inhibitor with specificity (HEK293: IC50 = 0.1 nM). Rapamycin has immunosuppressive activity and can induce autophagy.
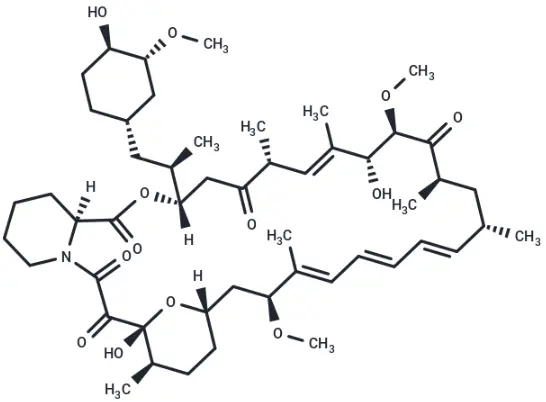
Molecular structure of Rapamycin
2. Background Introduction
mTOR (Mechanistic Target of Rapamycin) is a mammalian Rapamycin target protein, which belongs to the phosphatidylinositol-3-kinase-related protein kinase (PI3K-related kinase) family. It plays a central role in signal integration in cells, connecting nutrients, energy levels, growth factors and other environmental signals to regulate cell growth, protein synthesis, metabolism, proliferation and survival. mTOR mainly exists in the form of two functional complexes: mTORC1 and mTORC2.Among them, mTORC1 is highly sensitive to nutritional status and energy level, and is a key node in regulating autophagy and protein synthesis. The abnormal activation of mTOR pathway has important pathological significance in a variety of tumors, autoimmune diseases and metabolic diseases, so it has become an important target in drug development. [1]
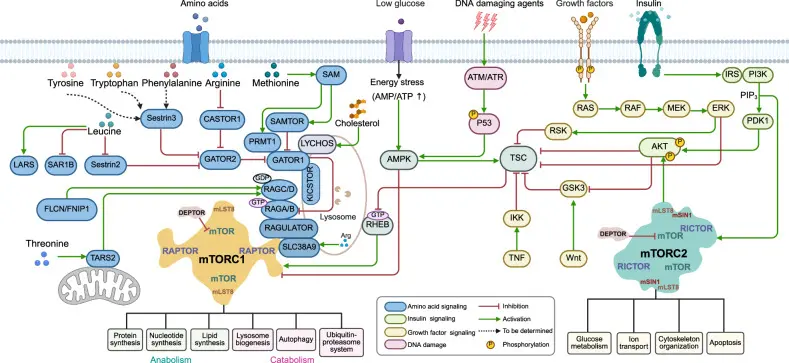
Overview of mTORC1 and mTORC2 signaling pathways [1]
Rapamycin is a natural compound that selectively inhibits mTOR protein kinase. Rapamycin first binds to the intracellular protein FKBP12 to form a complex, which specifically binds to and acutely inhibits the kinase activity of mTORC1, blocking the phosphorylation regulation of mTORC1 on its downstream effector molecules (such as S6 kinase 1,4E-BP1, ULK1, etc.), thereby reducing protein synthesis, regulating cell growth and autophagy. However, mTORC2 is less sensitive to Rapamycin and usually requires long-term drug exposure to indirectly interfere with its function, indicating that Rapamycin exerts its physiological effects mainly by inhibiting mTORC1. [2]
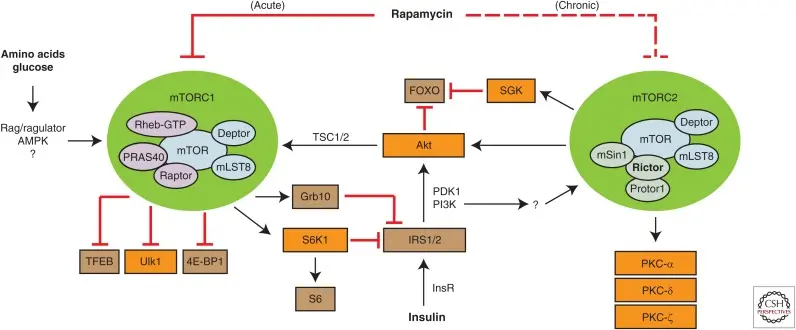
Mechanism target of Rapamycin (mTOR) signaling pathway [2]
3. Application References
Rapamycin inhibits B16 melanoma cell viability in vitro and in vivo by inducing autophagy and inhibiting the mTOR/p70‑S6k pathway

Research Overview:
In this study, B16 melanoma cells and mouse xenograft models were used to systematically evaluate the inhibitory effect of Rapamycin on melanoma growth in vitro and in vivo and its molecular mechanism. Through cell viability assay, colony formation assay and flow cytometry analysis, it was found that Rapamycin significantly reduced the proliferation and survival of B16 cells. At the same time, Western blot and other results showed that Rapamycin significantly inhibited the activation of mTOR/p70-S6K signaling pathway, reduced the phosphorylation level of downstream proteins, and up-regulated autophagy markers such as increased LC3-II, suggesting that Rapamycin participates in the anti-tumor process by inducing autophagy. In a mouse subcutaneous xenograft model, Rapamycin treatment significantly slowed tumor growth and reduced tumor volume. The overall results showed that Rapamycin significantly inhibited melanoma cells in vivo and in vitro models by inhibiting the mTOR / p70-S6K pathway and activating the autophagy pathway, providing an experimental basis for its potential application in melanoma treatment. [3]
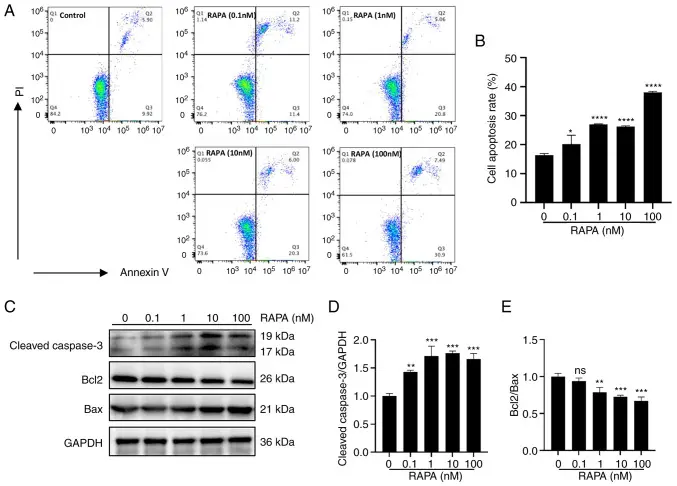
Rapamycin induced apoptosis of B16 cells in vitro [3]
CircABCA1 promotes ccRCC by reprogramming cholesterol metabolism and facilitating M2 macrophage polarization through IGF2BP3-mediated stabilization of SCARB1 mRNA

Research Overview:
In this study, clear cell renal cell carcinoma (ccRCC) was used to reveal the molecular mechanism of circular RNA CircABCA1 promoting tumor progression by reprogramming cholesterol metabolism and regulating tumor immune microenvironment. Studies have found that CircABCA1 is significantly up-regulated in ccRCC tissues, and functional experiments have shown that it can enhance tumor cell proliferation, migration and invasion. Mechanistically, CircABCA1 interacts with RNA binding protein IGF2BP3 to enhance its stability to SCARB1 mRNA, thereby up-regulating the expression of cholesterol uptake receptor SCARB1 and promoting cholesterol uptake and metabolic reprogramming in tumor cells. At the same time, metabolic changes further affect the tumor microenvironment, promote the polarization of macrophages to M2 phenotype, form an immunosuppressive environment and accelerate tumor progression. In vivo and in vitro experiments have confirmed that the intervention of CircABCA1 / IGF2BP3 / SCARB1 axis can significantly inhibit tumor growth. [4]
In this study, Rapamycin was used only as a pharmacological tool for mechanism verification experiments. By adding Rapamycin to inhibit mTOR signaling activity, we observed its effect on SCARB1-mediated cholesterol uptake, downstream metabolic reprogramming, and tumor cell phenotype, thus demonstrating that there is a functional association between the enhanced cholesterol metabolism caused by the CircABCA1 / IGF2BP3 / SCARB1 axis and the activation of the mTOR signaling pathway.
4. References
[1] Zhang H, Xiao X, Pan Z, Dokudovskaya S. mTOR signaling networks: mechanistic insights and translational frontiers in disease therapeutics. Signal Transduct Target Ther. 2025 Dec 30;10(1):428. doi: 10.1038/s41392-025-02493-4. PMID: 41469379; PMCID: PMC12753737.
[2] Lamming DW. Inhibition of the Mechanistic Target of Rapamycin (mTOR)-Rapamycin and Beyond. Cold Spring Harb Perspect Med. 2016 May 2;6(5):a025924. doi: 10.1101/cshperspect.a025924. PMID: 27048303; PMCID: PMC4852795.
[3] Wang P, Zhang H, Guo K, Liu C, Chen S, Pu B, Chen S, Feng T, Jiao H, Gao C. Rapamycin inhibits B16 melanoma cell viability in vitro and in vivo by inducing autophagy and inhibiting the mTOR/p70‑S6k pathway. Oncol Lett. 2024 Feb 2;27(4):140. doi: 10.3892/ol.2024.14273. PMID: 38385108; PMCID: PMC10877231.
[4] Ning H, Jiang Y, Li B, Ren J, Wang C, Wei L, Li L, Ran A, Li Z, Li J, Li W, Wang Y, Xiao B. CircABCA1 promotes ccRCC by reprogramming cholesterol metabolism and facilitating M2 macrophage polarization through IGF2BP3-mediated stabilization of SCARB1 mRNA. Mol Cancer. 2025 Jul 19;24(1):199. doi: 10.1186/s12943-025-02398-4. PMID: 40684174; PMCID: PMC12275331.
Other Articles
Subscription to TargetMol News
An essential round-up of science news, opinion and analysis, delivered to your inbox every weekday.

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.