Your shopping cart is currently empty
Your shopping cart is currently empty
TargetMol—Natural Product—Bafilomycin A1 (Cat. No. T6740, Cas. 88899-55-2), a Key Tool in the Study of Autophagic Flux
1. Product Introduction
Bafilomycin A1 (Cat. No. T6740, Cas. 88899-55-2), also known as Baf A1. Bafilomycin A1, a macrolide natural product, is a V-ATPase inhibitor (IC50 = 0.44 nM) with specificity. Bafilomycin A1 is an inhibitor of the late stage of autophagy, which blocks the fusion of autophagosomes and lysosomes. Bafilomycin A1 an cause lysosome‑dependent cell death at high concentrations.
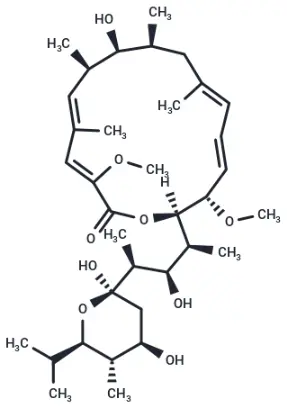
Molecular structure of Bafilomycin A1
2. Background Introduction
Vacuolar-type H+-ATPase (V-ATPase) is a kind of multi-subunit ATP-dependent proton pump widely existing in eukaryotic cells. It maintains the acidic environment of intracellular organelles such as lysosomes and endosomes by consuming ATP to drive proton (H+) transmembrane transport. This acidification is essential for the activation of proteases in lysosomes, the maturation and differentiation of endosomes and lysosomes, and the normal process of autophagic degradation. The proton pump activity of V-ATPase not only affects the endosomal / lysosomal pH, but also regulates key cellular processes such as protein degradation, receptor-ligand separation, and intracellular material transport and signal transduction. Its abnormal function is closely related to many disease states (such as neurodegenerative diseases and tumor cell homeostasis imbalance). [1]
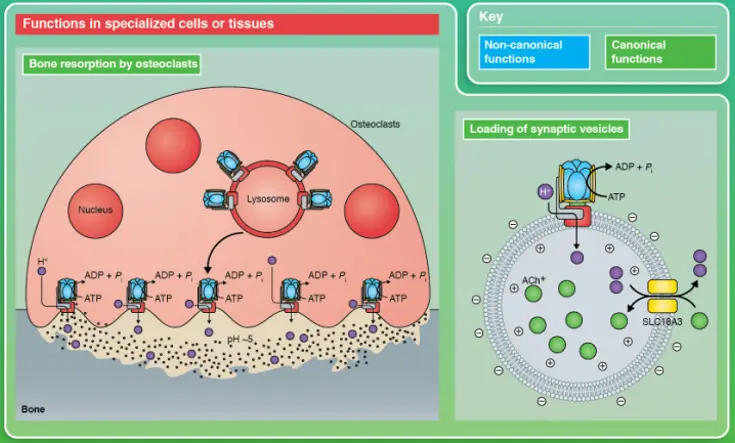
V-ATPase functions in specific cells or tissues [1]
Bafilomycin A1 is a macrolide natural product derived from Streptomyces, which is widely used as an efficient, specific and reversible inhibitor of V-ATPase in cell biology research. Its role is to target the V0 domain of the V-ATPase complex (especially the c-loop subunit), block V-ATPase-mediated proton transport, thereby inhibiting endosomal / lysosomal acidification. Since lysosomal acidic conditions are the key to multiple terminal steps of autophagy (including autophagosome-lysosomal fusion and autophagic flux maintenance), Bafilomycin A1 blocks these processes by inhibiting V-ATPase function, resulting in mature autophagosomes unable to fuse with lysosomes, resulting in autophagic flux termination and autophagosome accumulation. [2]
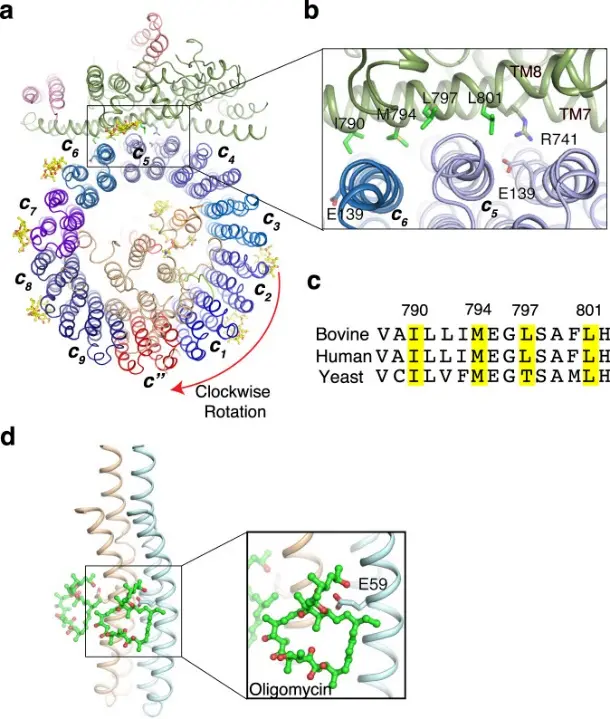
The mechanism of Bafilomycin A1 inhibiting c-ring rotation [2]
3. Application References
Bafilomycin A1 induces colon cancer cell death through impairment of the endolysosome system dependent on iron

Research Overview:
In this study, the effects of Bafilomycin A1 (BA1) on colon cancer and normal colon fibroblasts (CCD-18Co) cells were investigated. It was found that Bafilomycin A1 could selectively inhibit the survival and proliferation of various colon cancer cells at very low nanomolar concentrations, and cause lysosomal stress and intracellular iron homeostasis imbalance by destroying the late endosomal system (including abnormal number / size of lysosomes, changes in membrane permeability, and blocked autophagic flow), eventually leading to cancer cell death. In addition, iron (III) -citrate supplementation partially reversed these effects, indicating that iron deficiency caused by endolysosomal dysfunction is an important factor in Bafilomycin A1-mediated cell death, and this mechanism is related to endoplasmic reticulum stress and lipid droplet accumulation. Targeting endosomes and iron homeostasis is expected to be a new strategy for the treatment of colon cancer. [3]
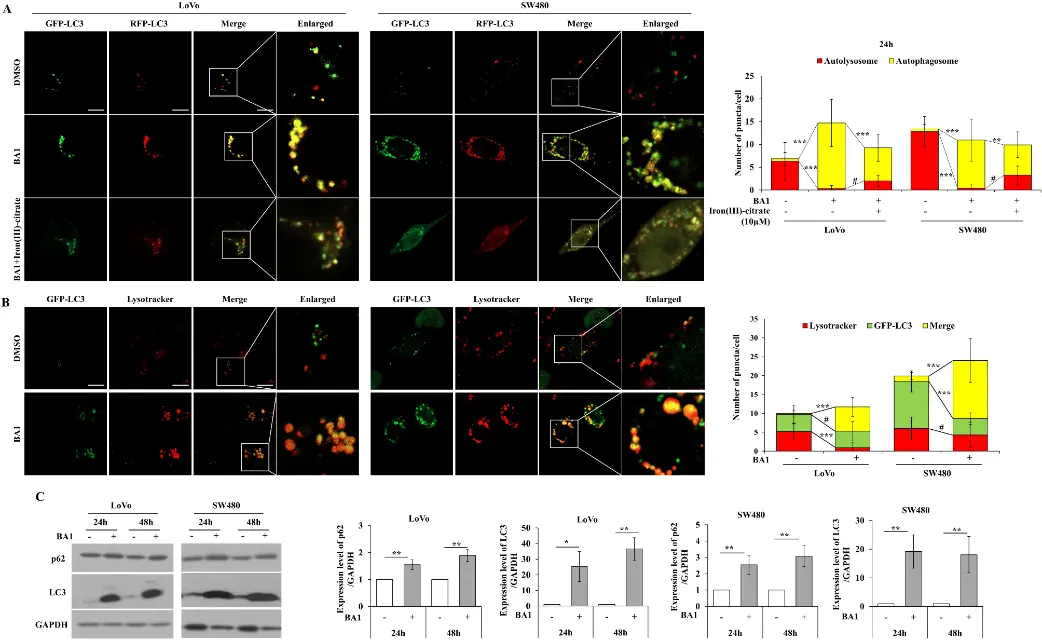
Bafilomycin A1 disrupted the endosomal system, including autophagy [3]
Osimertinib activates TFEB to trigger hepatocyte cytoplasmic vacuolation-associated cell death

Research Overview:
This study systematically revealed the molecular basis of Osimertinib-induced hepatocyte injury and unique cell death mechanism: Osimertinib can not only treat non-small cell lung cancer patients with EGFR mutations clinically, but also directly trigger obvious cytoplasmic vacuole formation in hepatocytes. Different from traditional apoptosis or necrosis, this vacuolization accompanied by cell death is caused by over-activated autophagy-lysosomal pathway. The molecular mechanism is that Osimertinib promotes the dephosphorylation and nuclear translocation of the transcription factor TFEB, activates TFEB-mediated lysosomal and autophagy gene expression, thereby enhancing autophagy-lysosomal biosynthesis, triggering the formation of a large number of lysosomes / autolysosomes and eventually leading to vacuolar accumulation and cell death. Studies have also shown that this cell damage can be alleviated by inhibiting the activation of TFEB (such as S-adenosyl-L-methionine), which provides a potential strategy for clinical control of Osimertinib hepatotoxicity and broadens the understanding of the mechanism of drug-induced liver injury. [4]
In this study, Bafilomycin A1 (Cat. No. T6740) was used as a classic lysosomal acidification inhibitor and late autophagy inhibitor to functionally verify whether Osimertinib-induced cell vacuolization is dependent on excessive activation of the autophagy-lysosomal pathway.
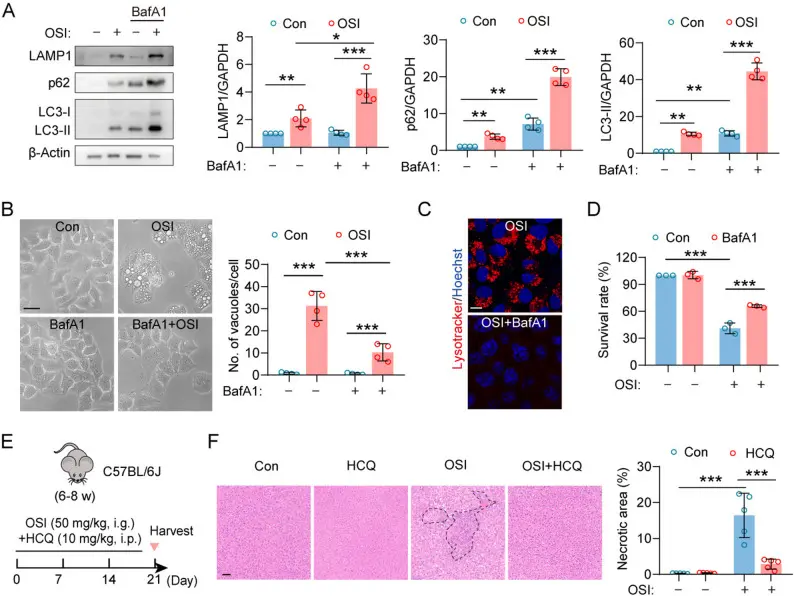
Hydroxychloroquine rescues Osimertinib-induced liver injury by inhibiting autophagy lysosomal pathway [4]
4. References
[1] Maxson ME, Grinstein S. The vacuolar-type H⁺-ATPase at a glance - more than a proton pump. J Cell Sci. 2014 Dec 1;127(Pt 23):4987-93. doi: 10.1242/jcs.158550. PMID: 25453113.
[2] Wang R, Wang J, Hassan A, Lee CH, Xie XS, Li X. Molecular basis of V-ATPase inhibition by bafilomycin A1. Nat Commun. 2021 Mar 19;12(1):1782. doi: 10.1038/s41467-021-22111-5. PMID: 33741963; PMCID: PMC7979754.
[3] Min DH, Kim D, Hong ST, Kim J, Kim MJ, Kwon SH, Kim A, Lee JY. Bafilomycin A1 induces colon cancer cell death through impairment of the endolysosome system dependent on iron. Sci Rep. 2025 Feb 12;15(1):5148. doi: 10.1038/s41598-025-89127-5. PMID: 39934167; PMCID: PMC11814099.
[4] Qiu Y, Liu Y, Ding H, Xin W, Lu S, Hu Y, Chen J, Weng Q, Wang J, Fang L. Osimertinib activates TFEB to trigger hepatocyte cytoplasmic vacuolation-associated cell death. Cell Commun Signal. 2026 Jan 30;24(1):142. doi: 10.1186/s12964-026-02688-4. PMID: 41612351; PMCID: PMC12934022.
Other Articles
Subscription to TargetMol News
An essential round-up of science news, opinion and analysis, delivered to your inbox every weekday.

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.