Your shopping cart is currently empty
Your shopping cart is currently empty
TargetMol—Inhibitor—RSL3 (Cat. No. T3646,CAS. 1219810-16-8), Inhibition of GPX4 induces ferroptosis
1. Product Introduction
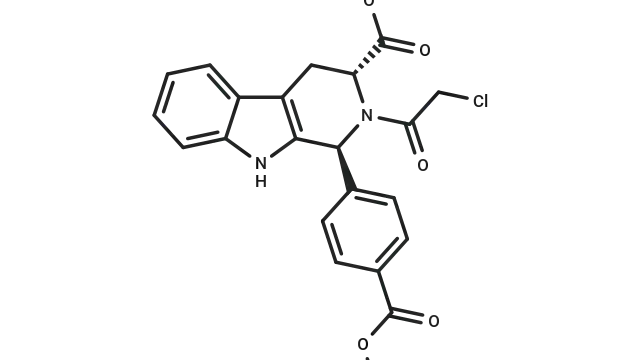
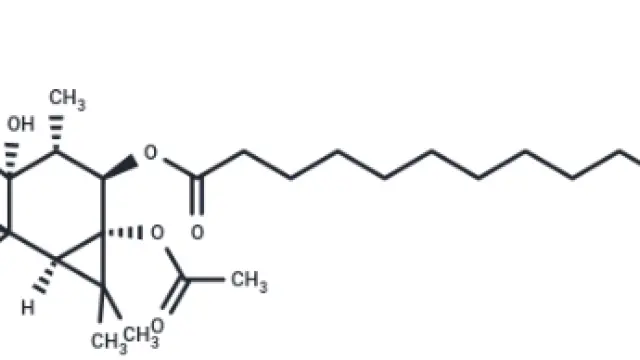
RSL3 (Cat. No. T3646,CAS. 1219810-16-8), also known as RSL3 1S,1S,3R-RSL3. RSL3 (RSL3 1S) is a GPX4 directly inhibits GPX4 (IC₅₀ = 100 nM) and does NOT target system xc- that blocks GSH synthesis. RSL3 is a VDAC-independent ferroptosis activator that is selective for tumor cells carrying oncogenic RAS.
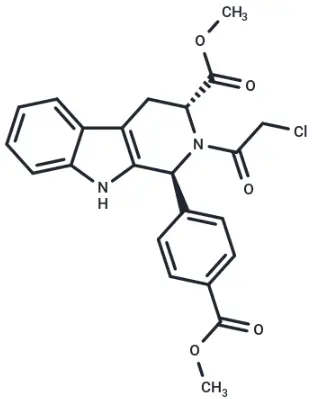
Fig. Molecular structure of RSL3
2. Background Introduction
Ferroptosis is an iron-dependent form of regulated cell death, which is different from traditional cell death types such as apoptosis and necrosis. Its core feature is the accumulation of lipid peroxides on the cell membrane. In this process, glutathione peroxidase 4 (GPX4) and cystine / glutamate exchange transporter (system xc-) are two key regulatory nodes. GPX4 is a selenium-containing glutathione-dependent peroxidase, which can reduce lipid peroxides to corresponding alcohols, thereby preventing the expansion of lipid free radicals and cell membrane damage. Its normal function is essential for inhibiting ferroptosis. Studies have shown that impaired GPX4 activity can lead to the accumulation of lipid reactive oxygen species (ROS) and trigger ferroptosis, which is characterized by rapid accumulation of oxidized lipids and iron-dependent cell death. Different from this, system xc- is a cystine/glutamate reverse transporter located on the cell membrane. Its main function is to maintain intracellular GSH (glutathione) levels by ingesting cystine, and GSH is an essential cofactor for GPX4 to perform antioxidant functions. Therefore, blocking system xc- can reduce intracellular cystine and GSH synthesis, thereby indirectly weakening the function of GPX4 and promoting ferroptosis. Based on this, inhibition of system xc- or direct inhibition of GPX4 is considered to be an effective strategy to induce ferroptosis. [1]
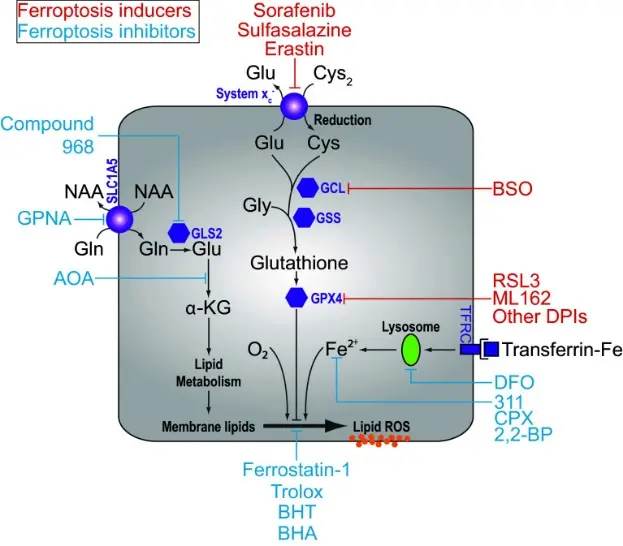
Overview of the ferroptosis pathway [1]
RSL3 is a small molecule ferroptosis inducer that has been found to selectively kill cell lines carrying carcinogenic RAS mutations for the first time in high-throughput screening. Unlike ferroptosis inducers (such as erastin) that lead to GSH deficiency by inhibiting system xc, RSL3 inhibits its catalytic activity by directly interacting with GPX4 to trigger ferroptosis. Chemical probe and affinity mass spectrometry studies have shown that the active isomer of RSL3 can covalently bind to the active site of GPX4, making it lose the ability to reduce lipid peroxides, thereby promoting the accumulation of lipid peroxides and ferroptosis. This mechanism allows RSL3 to effectively reduce the survival rate of sensitive cells at the nanomolar level and show relative selectivity to tumor cells. In addition, RSL3 does not significantly affect the GSH level itself, so it is classified as ' Class II ferroptosis inducer (direct GPX4 inhibitor) ', which is distinguished from ' Class I ferroptosis inducer ' that inactivates GPX4 indirectly through GSH deficiency. [2]
3. Application References
RSL3 Drives Ferroptosis Through GPX4 Inactivation and ROS Production in Colorectal Cancer

Research Overview:
This study systematically evaluated the anti-tumor effect of RSL3 and its molecular mechanism in a variety of human colorectal cancer cell lines (HCT116, HT29, LoVo). The results showed that RSL3 treatment significantly reduced cell viability, and increased intracellular ROS and lipid peroxidation levels in a time-and dose-dependent manner, accompanied by impaired GPX4 protein function. By adding ferroptosis inhibitor Ferrostatin-1, iron chelating agent and GPX4 overexpression, the study further verified that RSL3-induced cell death mainly depended on ferroptosis pathway rather than apoptosis or necrosis. In addition, RSL3 treatment also caused mitochondrial morphological changes and membrane lipid oxidative damage. This study confirmed that RSL3 drives iron-dependent programmed cell death in colorectal cancer cells by inhibiting GPX4 activity and promoting ROS and lipid peroxidation accumulation from the molecular, cellular and functional levels, which provides an experimental basis for the use of ferroptosis mechanism to carry out colorectal cancer treatment strategies. [3]
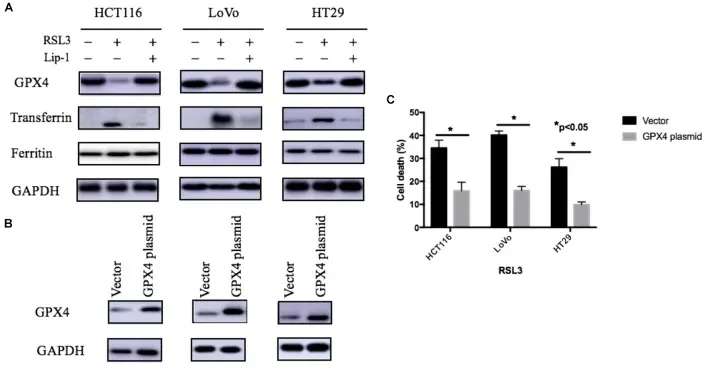
The expression of iron regulatory protein and the effect of GPX4 on RSL-3-induced cell death through ferroptosis [3]
Identification of a targeted ACSL4 inhibitor to treat ferroptosis-related diseases

Research Overview:
In this study, a small molecule compound AS-252424 (AS) was identified by screening the kinase inhibitor library, and it was found that it could specifically bind to the Gln464 site of the fatty acid metabolic enzyme ACSL4 and inhibit its enzyme activity, thereby significantly inhibiting intracellular lipid peroxidation and ferroptosis. It effectively blocked RSL3-induced ferroptosis in an in vitro human-mouse cell model. At the same time, nanoparticle carriers were used to alleviate ferroptosis-related organ damage in mouse renal ischemia-reperfusion injury and acute liver injury models, indicating that AS, as a specific ferroptosis inhibitor for ACSL4, has potential therapeutic value as a ferroptosis-related disease. [4]
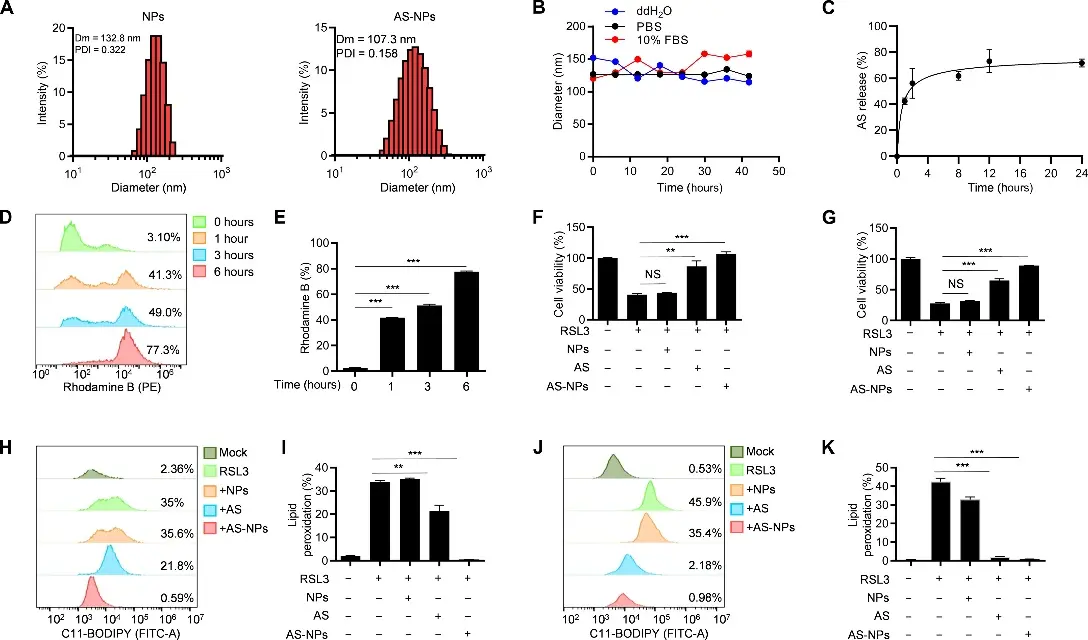
AS-NPs inhibit ferroptosis [4]
4. References
[1] Cao JY, Dixon SJ. Mechanisms of ferroptosis. Cell Mol Life Sci. 2016 Jun;73(11-12):2195-209. doi: 10.1007/s00018-016-2194-1. Epub 2016 Apr 5. PMID: 27048822; PMCID: PMC4887533.
[2] Stockwell BR. Ferroptosis turns 10: Emerging mechanisms, physiological functions, and therapeutic applications. Cell. 2022 Jul 7;185(14):2401-2421. doi: 10.1016/j.cell.2022.06.003. PMID: 35803244; PMCID: PMC9273022.
[3] Sui X, Zhang R, Liu S, et al. RSL3 Drives Ferroptosis Through GPX4 Inactivation and ROS Production in Colorectal Cancer. Front Pharmacol. 2018 Nov 22;9:1371. doi: 10.3389/fphar.2018.01371. PMID: 30524291; PMCID: PMC6262051.
[4] Huang Q, Ru Y, Luo Y, et al. Identification of a targeted ACSL4 inhibitor to treat ferroptosis-related diseases. Sci Adv. 2024 Mar 29;10(13):eadk1200. doi: 10.1126/sciadv.adk1200. Epub 2024 Mar 29. PMID: 38552012; PMCID: PMC10980261.
Other Articles
Subscription to TargetMol News
An essential round-up of science news, opinion and analysis, delivered to your inbox every weekday.

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.