Your shopping cart is currently empty
Your shopping cart is currently empty
TargetMol—Disease Modeling—Cisplatin (Cat. No. T1564, CAS. 15663-27-1), Regulating Injury, Ferroptosis and Autophagy
1. Product Introduction
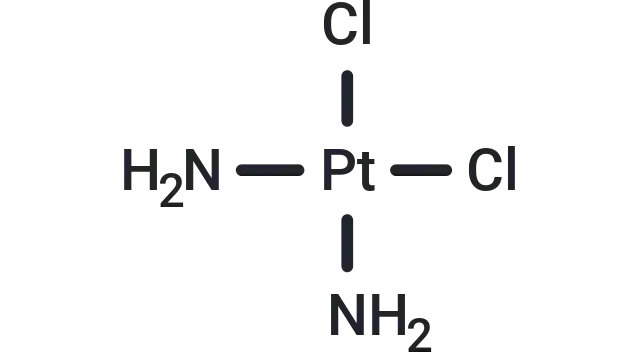
Cisplatin (Cat. No. T1564, CAS. 15663-27-1), also known as cis-Diaminodichloroplatinum, CDDP. Cisplatin (CDDP) is a chemotherapeutic drug with anti-tumor activity. It is a typical DNA cross-linking agent that can inhibit DNA synthesis and cause DNA damage by forming DNA adducts in cancer cells, leading to cell death. In addition, Cisplatin can also activate ferroptosis and induce autophagy. In animal experiments, Cisplatin is often used to construct chronic kidney injury and acute renal failure models.
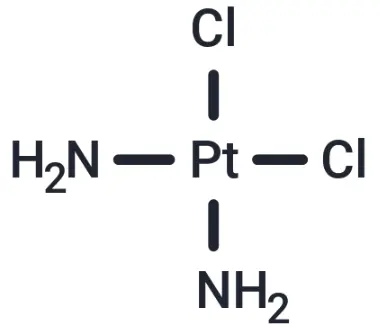
Molecular structure of Cisplatin
2. Background Introduction
DNA is one of the core targets of most chemotherapeutic drugs, especially in tumor cells with active proliferation. DNA damage can inhibit tumor growth by forming adducts or cross-linked structures, blocking replication and transcription processes, and activating cell cycle arrest and programmed cell death signaling pathways. Recent studies have further shown that in addition to the classical DNA damage response, non-apoptotic cell death pathwayssuch as ferroptosis and autophagy are also involved in the regulation of tumor cell fate. Ferroptosis is a regulated cell death driven by iron-dependent lipid peroxidation, which is characterized by glutathione (GSH) depletion and glutathione peroxidase 4 (GPX4) inactivation. At the same time, autophagy, as an important mechanism of cell stress response, plays a key role in maintaining cell homeostasis and regulating cell survival-death balance. The discovery of these novel cell death pathways provides a broader perspective for the study of tumor therapeutic targets based on DNA damage. [1]
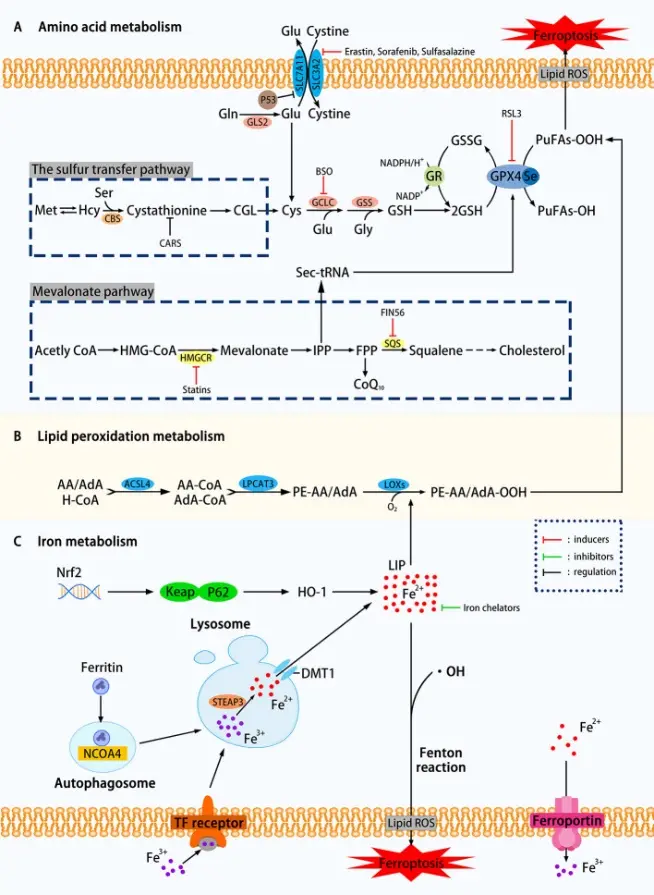
Mechanism of cell ferroptosis [1]
Cisplatin is one of the most widely used platinum-based anti-tumor drugs in clinical practice. Its classical mechanism of action is mainly to form DNA-platinum adducts and intra-chain/inter-chain cross-linking structures by binding to guanine bases in DNA after entering cells, thereby interfering with DNA replication and transcription. The process eventually triggers cell cycle arrest and induces apoptosis. This DNA damage response is the core basis of its anti-tumor activity. In addition to the classic DNA damage pathway, recent studies have found that Cisplatin can also play a role through a variety of non-apoptotic pathways, such as inducing ferroptosis-related lipid peroxidation and regulating autophagy, thereby further enhancing its cytotoxic effects. [2]
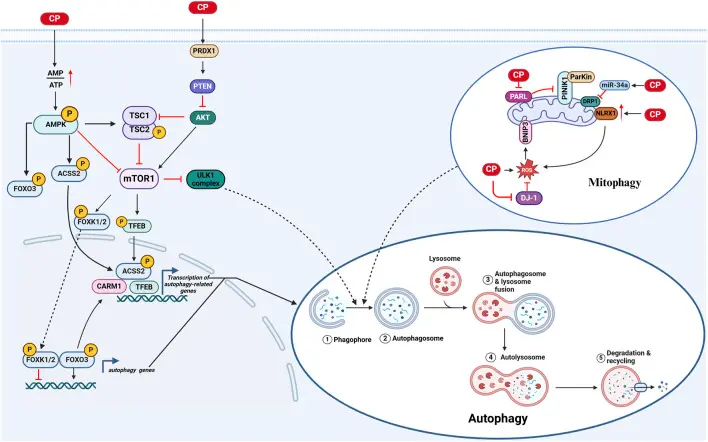
Autophagy in CIO (cisplatin-induced ototoxicity) [2]
3. Application References
Tetrahedral DNA Nanostructures Inhibit Ferroptosis and Apoptosis in Cisplatin-induced Renal Injury

Research Overview:
This study focused on cisplatin-induced acute kidney injury (AKI) and its cell death mechanism, and explored the role and mechanism of tetrahedral DNA nanostructures (TDNs) in renal protection. In this study, a mouse model of cisplatin-induced acute kidney injury (AKI) was established, and the mechanism of renal tubular cell injury was systematically evaluated in vitro and in vivo. It was found that TDNs could reduce the production of lipid reactive oxygen species (ROS) and restore the down-regulation of glutathione peroxidase 4 (GPX4), thereby inhibiting ferroptosisinduced by RSL3. In addition, studies have shown that TDNs attenuate cisplatin-induced ferroptosis by reversing the down-regulation of GPX4 and attenuating cisplatin-induced apoptosis by reducing the cleavage of poly (ADP-ribose) polymerase (PARP). [3]
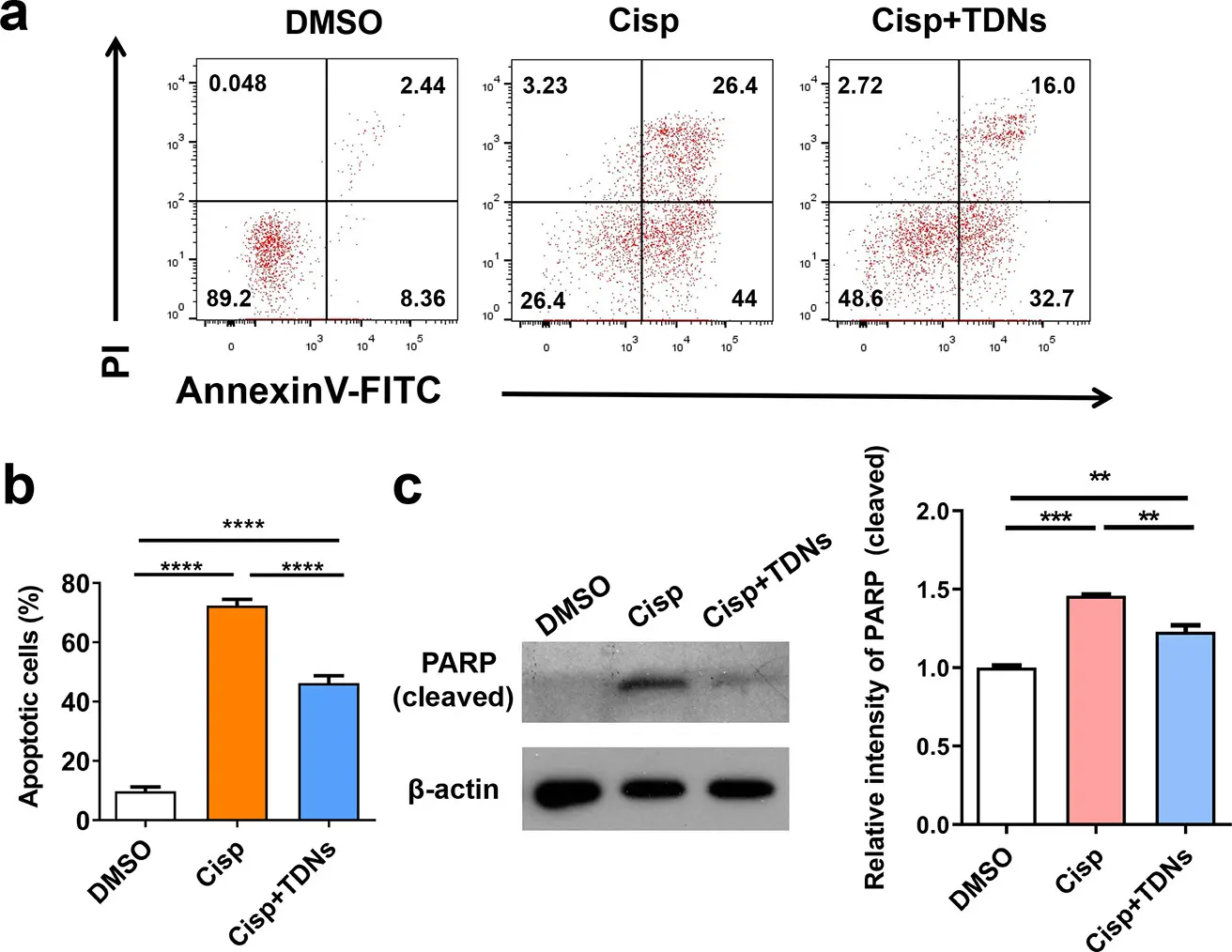
TDN inhibits cisplatin-induced apoptosis [3]
Mitophagy alleviates cisplatin-induced renal tubular epithelial cell ferroptosis through ROS/HO-1/GPX4 axis

Research Overview:
This study investigated the mechanism of BNIP3-mediated and PINK1-PARK2-mediated mitophagy-induced ferroptosis in cisplatin-induced acute kidney injury. In vivo and in vitro experimental models have shown that cisplatin treatment can significantly induce renal tubular epithelial cell damage, manifested as a large accumulation of ROS, enhanced lipid peroxidation, iron homeostasis disorder, and down-regulation of GPX4 expression, thereby triggering ferroptosis. At the same time, studies have found that cisplatin can also cause mitochondrial dysfunction, and activation of mitophagy can selectively remove damaged mitochondria, reduce ROS production and alleviate oxidative stress. This study elucidates a new mechanism by which BNIP3-mediated and PINK1-PARK2-mediated mitophagy protects cisplatin-induced renal tubular epithelial cell ferroptosis through the ROS/HO1/GPX4 axis. [4]
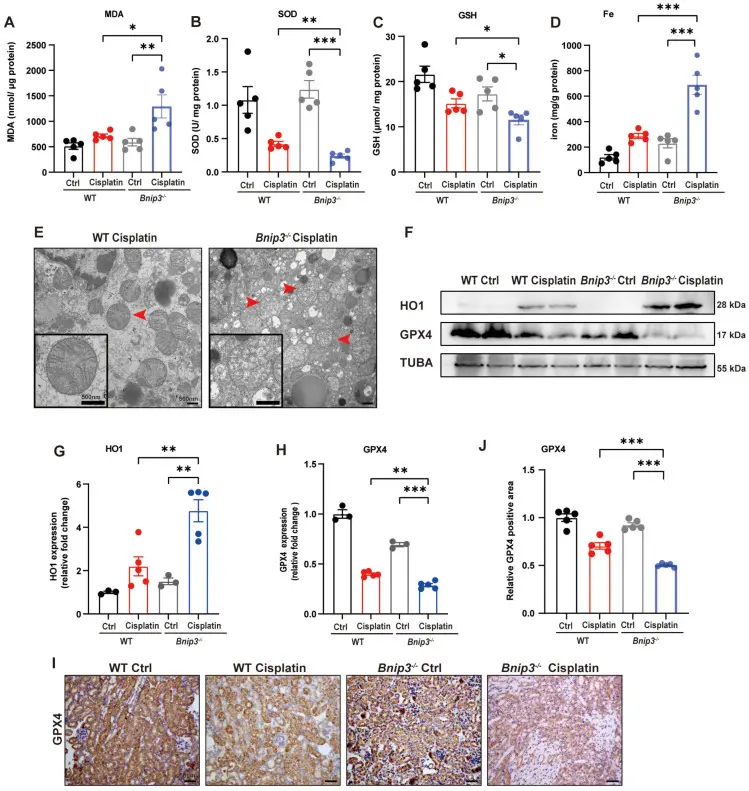
Cisplatin-induced ROS, lipid peroxidation and RTEC ferroptosis were increased in Bnip3 knockout kidneys. [4]
Targeting the ATM-TGS1-BRCA1 Axis Overcomes Genotoxic Therapy Resistance in Pancreatic Adenocarcinoma

This study reveals a key molecular mechanism for the resistance of pancreatic ductal adenocarcinoma to genotoxic therapy, that is, the core regulatory role of the ATM-TGS1-BRCA1 signaling axis in DNA damage repair. The authors found that in the process of DNA damage response, ATM kinase promotes the maintenance of BRCA1 function by regulating TGS1 activity, thereby enhancing the ability of homologous recombination repair, and making tumor cells more tolerant to genotoxic treatments such as radiotherapy and chemotherapy. Inhibition of TGS1 or interference with the signal axis can significantly weaken the BRCA1-mediated DNA repair ability, leading to the accumulation of DNA damage, thereby restoring the sensitivity of tumor cells to genotoxic therapy. This study suggests that targeting the ATM-TGS1-BRCA1 axis to destroy the DNA repair advantages of tumor cells is a potential new strategy to overcome pancreatic cancer treatment resistance and enhance the efficacy of radiotherapy and chemotherapy. [5]
In this study, cisplatin was used as a representative DNA damage inducer for genotoxic therapy. The authors used cisplatin to artificially create DNA double-strand damage in in vitro and in vivo models to evaluate the role of the ATM-TGS1-BRCA1 signaling axis in DNA damage repair and therapeutic resistance. By comparing the response of tumor cells to cisplatin treatment before and after the signal axis was inhibited or interfered, it was proved that this pathway enhanced the ability of homologous recombination repair, resulting in tolerance to cisplatin. Therefore, cisplatin is mainly used as a functional experimental tool drug in this paper to verify how the molecular axis mediates the resistance mechanism of pancreatic cancer to DNA damage treatment.
4. References
[1] Lin X, Ping J, Wen Y, et al. The Mechanism of Ferroptosis and Applications in Tumor Treatment. Front Pharmacol. 2020 Jul 22;11:1061. doi: 10.3389/fphar.2020.01061. PMID: 32774303; PMCID: PMC7388725.
[2] Dai D, Chen C, Lu C, et al. Apoptosis, autophagy, ferroptosis, and pyroptosis in cisplatin-induced ototoxicity and protective agents. Front Pharmacol. 2024 Sep 24;15:1430469. doi: 10.3389/fphar.2024.1430469. PMID: 39380912; PMCID: PMC11459463.
[3] Li J, Wei L, Zhang Y, et al. Tetrahedral DNA Nanostructures Inhibit Ferroptosis and Apoptosis in Cisplatin-induced Renal Injury. ACS Appl Bio Mater. 2021 Jun 21;4(6):5026-5032. doi: 10.1021/acsabm.1c00294. Epub 2021 May 25. PMID: 35007051.
[4] Lin Q, Li S, Jin H, et al. Mitophagy alleviates cisplatin-induced renal tubular epithelial cell ferroptosis through ROS/HO-1/GPX4 axis. Int J Biol Sci. 2023 Feb 13;19(4):1192-1210. doi: 10.7150/ijbs.80775. PMID: 36923942; PMCID: PMC10008689.
[5] Li C, Zhao X, Li X, Wang C, Huo Z, Xu X, Kang W, Nowsheen S, Aziz K, Sun G, Liu Z, Lou Z, Deng M. Targeting the ATM-TGS1-BRCA1 Axis Overcomes Genotoxic Therapy Resistance in Pancreatic Adenocarcinoma. Cancer Res. 2026 Feb 2;86(3):730-745. doi: 10.1158/0008-5472.CAN-25-1435. PMID: 41183146.
Other Articles
Subscription to TargetMol News
An essential round-up of science news, opinion and analysis, delivered to your inbox every weekday.

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.