| Disease Modeling Protocol | Hyperuricemia model- Modeling Mechanism:
Adenine, as a purine precursor, increases uric acid production and synergistically raises serum uric acid (UA) levels with potassium oxalate. Persistent hyperuricemia leads to dysfunction of renal uric acid transporters (upregulation of reabsorption transporters and downregulation of secretory transporters), which in turn causes renal tubular damage and inflammatory infiltration, mimicking the pathophysiological process of human hyperuricemia and related kidney damage. - Related Products:
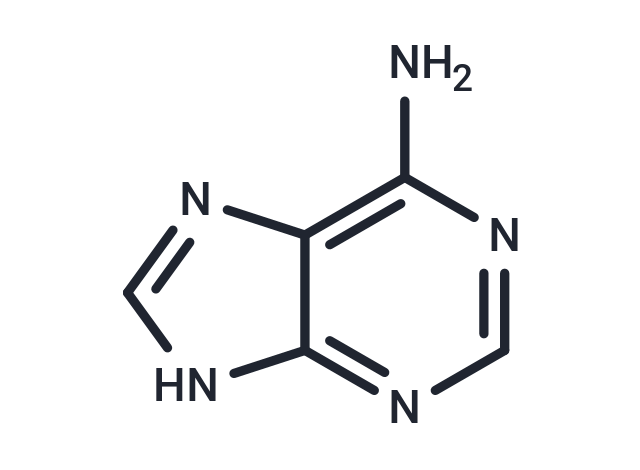
Adenine (T0064) - Modeling Method:
Experimental Subject: Mouse, Kunming mouse, male, 7 weeks old, weight about 20-25g Dosage and Administration Route: ① Core modeling: potassium oxalate (200 mg/kg)+adenine (50 mg/kg) suspended in 5% gum arabic solution, administered by gavage;
② Control treatment: equal volume of 5% gum arabic solution administered by gavage in the same manner;
③ Intervention verification (optional): benzbromarone (50 mg/kg) suspended in 5% gum arabic solution, administered by gavage, given 1 hour apart from the modeling drug. Dosing Frequency and Duration Model: once daily for 21 consecutive days - Validation:
Serum uric acid (UA): Significantly increased on day 3 of modeling (model group 123.45 μmol/L vs control group 42.03 μmol/L, p<0.001), peaking on day 7 (approximately 175.70 μmol/L, 3 times that of the control group), and then remained stable at a high level; Uric acid clearance (Cur): Significantly decreased from day 3 (model group 0.73 vs control group 1.56), and continued to decrease with the extension of modeling time (decreased to 0.19 on day 21); Urine indicators: The 24-hour urine volume of the model group was significantly increased, but the urinary uric acid excretion was reduced; Pathological indicators: - HE staining: Mild renal tubular epithelial cell necrosis and inflammatory infiltration appeared on day 3; the damage worsened from day 10, manifested as renal tubular dilation, blurred cell boundaries, and a large number of inflammatory cell infiltrations, with amyloid bodies visible in some renal tubules; Molecular indicators: - Renal transporters: Western blot detection showed that on day 3 of modeling... From day 7 onwards, the expression of URAT1 and GLUT9 (uric acid reabsorption transporters) proteins was significantly upregulated, while the expression of ABCG2 and OAT1 (uric acid secretion transporters) was significantly downregulated. NPT1 (secretion transporter) was significantly downregulated from day 7, with the most significant change on day 10. General condition: - Mice in the model group were lethargic, had decreased weight, dry fur, increased water intake and decreased food intake, which were significantly different from the normal group.
*Precautions: On days 3, 7, 10, 14, 17, and 21 after blood collection, these mice were subsequently euthanized by carbon dioxide. *References:Wen S,et,al. The Time-Feature of Uric Acid Excretion in Hyperuricemia Mice Induced by Potassium Oxonate and Adenine. Int J Mol Sci. 2020 Jul 22;21(15):5178. Hyperuricemic nephropathy model- Modeling Mechanism:
① Adenine, as a purine precursor, increases uric acid production, and excessive uric acid deposition in the kidneys causes crystalline damage; ② Ethambutol hydrochloride inhibits renal uric acid excretion, exacerbating uric acid accumulation in the body and further aggravating kidney damage; ③ The two work together to activate inflammatory pathways, leading to upregulation of pro-inflammatory factors such as IL-6 and TNF, triggering renal tissue inflammation, glomerular sclerosis, and interstitial fibrosis, mimicking the core pathological process of human hyperuricemic nephropathy. - Related Products:
Adenine (T0064) - Modeling Method:
Experimental Subject: Rat, Wistar, Male, 7–8 weeks old, weighing 200–220 g Dosage and Administration Route: ① Core modelling: Adenine (100 mg/kg)+Ethambutol hydrochloride (250 mg/kg), Dissolved in deionised water to prepare a 1% adenine+2.5% ethambutol hydrochloride suspension, Administered via gastric lavage;
② Control treatment: Equal volume of deionised water, Administered via gastric lavage using the same method;
③ Intervention validation (optional):
- Allopurinol (50 mg/kg), 0.5% solution administered via gastric lavage, commencing from week 2 of modelling, for 2 consecutive weeks;
- Chicory formula (CF): high dose 8.64 g/kg, low dose 2.16 g/kg; administered via gastric lavage as a decoction; commencing from week 2 of modelling, for 2 consecutive weeks. Dosing Frequency and Duration Model: Once daily, for 3 consecutive weeks - Validation:
Key indicators (serum biochemistry): - Serum uric acid (UA): The model group reached 2.40 μMol/L, significantly higher than the control group (2.25 μMol/L). After intervention with allopurinol and high-dose CF, it decreased to 1.57 μMol/L (p<0.05); - Renal function indicators: Serum urea (UREA) increased from 6.75 mMol/L to 15.87 mMol/L, and serum creatinine (CREA) increased from 38.5 μmol/L to 64.83 μMol/L (p<0.001), both significantly decreasing after intervention; Pathological indicators: - HE staining showed disordered kidney structure in the model group, with glomerular sclerosis, renal tubular epithelial cell damage, renal interstitial inflammation, and uric acid crystal deposition. Pathological damage was significantly reduced in the intervention group; Molecular indicators: - Significantly upregulated expression of IL-6, TP53, and TNF mRNA in renal tissue (p<0.05), and VEGFA and CASP3 mRNA expression... The expression was downgraded, and the trend could be reversed after intervention.
*Precautions: To alleviate pain, rats were euthanized by intraperitoneal injection of sodium pentobarbital. *References:Li N,et,al. Integration of network pharmacology and intestinal flora to investigate the mechanism of action of Chinese herbal Cichorium intybus formula in attenuating adenine and ethambutol hydrochloride-induced hyperuricemic nephropathy in rats. Pharm Biol. 2022 Dec;60(1):2338-2354. |
 Your shopping cart is currently empty
Your shopping cart is currently empty

![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.![]() stock solution to 400 μL PEG300
stock solution to 400 μL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.