Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
Synonyms: ENT 38
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 5 g | $40 | - | In Stock | |
| 1 mL x 10 mM (in DMSO) | $29 | In Stock | In Stock |
| Description | Phenothiazine (ENT 38) is a class of agents exhibiting antiemetic, antipsychotic, antihistaminic, and anticholinergic activities. Phenothiazines antagonize the dopamine D2-receptor in the chemoreceptor trigger zone (CTZ) of the brain, potentially preventing chemotherapy-induced emesis. |
| In vitro | Phenothiazines mostly substitutes at position 10 with the dialkylaminoalkyl groups and additionally at position 2 with small groups exhibit valuable activities such as neuroleptic, antiemetic, antihistaminic, antipuritic, analgesic and antihelmintic. 2-trifluoromethyl-10-(4-aminobutyl)phenothiazine inhibits S. cerevisiae strains and T. mentagrophites with MIC of 0.4 μg/mL and 1.5 μg/mL, respectively. 10-carbamoylalkylphenothiazines shows significant activities against Gram-positive Bacillus subtilis with MIC's in the range of 7.8 μg/mL–30 μg/mL. The tetracyclic phenothiazines (modified with the naphthoquinone ring) shows significant actibacterial activity against S. aureus with the MIC50 of 12.5 μg/mL. Phenothiazines with the butylene linker are more effective than with the propylene linker, the 2-chloro-10-chloroethylureidobutyl derivative giving GI50 of 1.4 μM and 1.6 μM against 4 leukemia cell lines and 7 colon cancer cell lines. 10-Amino(hydroxy)propylphenothiazines (5 μM) induces a marked G2/M phase of cell-cycle arrest followed by cell death in human transformed WI38VA cells after 2-day incubation. [1] Phenothiazine drugs undergo extensive metabolism in the body before being excreted, mainly ring hydroxylation, ring sulphoxidation, N-demethylation, N-oxidation, sulphate and glucuronide conjugation. Phenothiazines have considerably lower binding affinities to α2-adrenoceptors than to dopamine D2 receptors and al-adrenoceptors. [2] Phenothiazines have significant in vitro activity against susceptible, polydrug- and multidrug-resistant strains of M. tuberculosis, as well as enhancing the activity of some agents employed for first-line treatment. [3] |
| Synonyms | ENT 38 |
| Molecular Weight | 199.27 |
| Formula | C12H9NS |
| Cas No. | 92-84-2 |
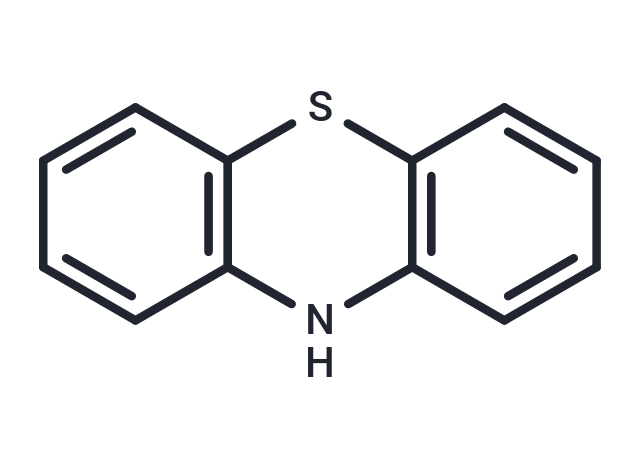
| Smiles | N1C2=C(SC3=C1C=CC=C3)C=CC=C2 |
| Relative Density. | 1.296. Temperature:20 °C. |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. | |||||||||||||||||||||||||||||||||||
| Solubility Information | DMSO: 247.5 mg/mL (1242.03 mM), Sonication is recommended. Ethanol: < 1 mg/mL (insoluble or slightly soluble) H2O: < 1 mg/mL (insoluble or slightly soluble) | |||||||||||||||||||||||||||||||||||
| In Vivo Formulation | 10% DMSO+40% PEG300+5% Tween 80+45% Saline: 2 mg/mL (10.04 mM), Sonication is recommended. Please add the solvents sequentially, clarifying the solution as much as possible before adding the next one. Dissolve by heating and/or sonication if necessary. Working solution is recommended to be prepared and used immediately. The formulation provided above is for reference purposes only. In vivo formulations may vary and should be modified based on specific experimental conditions. | |||||||||||||||||||||||||||||||||||
Solution Preparation Table | ||||||||||||||||||||||||||||||||||||
DMSO
Note : The dilution table applies only to solid products. For liquid products, please calculate the stock solution based on the stated concentration and/or density. | ||||||||||||||||||||||||||||||||||||
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 μL PEG300
stock solution to 400 μL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 μL Tween 80 and mix well until fully clarified.
3) Add 450 μL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.