 Your shopping cart is currently empty
Your shopping cart is currently empty

Synonyms: Methyl ferulate, Methyl 4'-hydroxy-3'-methoxycinnamate
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 1 g | $31 | - | In Stock | |
| 5 g | $49 | - | In Stock | |
| 10 g | $66 | - | In Stock | |
| 1 mL x 10 mM (in DMSO) | $29 | - | In Stock |
| Description | FERULIC ACID METHYL ESTER (Methyl ferulate) is a hydroxycinnamic acid that is abundant in plants and originally derived from giant fennel (F. communis). This naturally-occurring phenolic has antioxidant activities that provide protection against inflammation and cancer. Ferulic acid methyl ester is a lipophilic derivative of ferulic acid, demonstrating increased ability to cross cell membranes. |
| In vitro | Ferulic acid methyl ester has less antioxidant capacity than ferulic acid in neuronal PC12 cells (IC50 = 74.7 μM for ferulic acid ethyl ester vs. 44.6 μM for ferulic acid, 2,2-diphenyl-1-picrylhydrazyl radical scavenging). Ferulic acid methyl ester, at 10-25 μg/ml, inhibits the release of pro-inflammatory cytokines, blocks the expression of COX-2, and reduces nitric oxide generation from LPS-stimulated macrophages. |
| Synonyms | Methyl ferulate, Methyl 4'-hydroxy-3'-methoxycinnamate |
| Kinase Assay | DPPH radical scavenging activity of caffeic acid and ferulic acid derivatives was assessed as previously described withsome modifications. Briefly, the test compound was dissolved in dimethyl sulfoxide and 4 different concentrations weremixed with a methanolic solution of DPPH 100 mM in duplicate. After 30 min of incubation at room temperature in the dark, theabsorbance at 517 nm was measured by a spectrophotometer . The concentrations (in the range 1-100 μM) were carefully chosen for each compound in order to produce a suitable doseeresponse curve. The percent inhibition of the radical was calculated based on the absorbance of the mixture compared to the absorbance of DPPH solution alone. |
| Cell Research | Hydrogen peroxide (8.8 M solution) stored at 4 C was firstdiluted in PBS to prepare a 100 mM solution on the day of theexperiment. This was further diluted in growth medium to preparethe final working solution. PC12 cells were plated in collagencoated96-well microplates at a density of 5 105 cells/ml(100 μl per well). Blank wells contained only growth medium forbackground correction. After 48 h of incubation to allow for cellattachment, 20 μl of growth medium supplemented with differentconcentrations of HCAs were added in triplicate wells and preincubatedfor 1 h. Maximum concentration of DMSO in the wellswas kept below 0.2%. Afterwards, 20 μl of H2O2 solution was added.The concentration of H2O2 in the well was 75 μM. After anotherhour, the medium was replaced with fresh one and cells wereincubated overnight. In the end, the medium was replaced with30 μl of MTT 0.5 mg/ml dissolved in RPMI without phenol red.Formazan crystals were solubilised in 200 μl DMSO after 1.5 h ofincubation at 37 C. |
| Molecular Weight | 208.21 |
| Formula | C11H12O4 |
| Cas No. | 2309-07-1 |
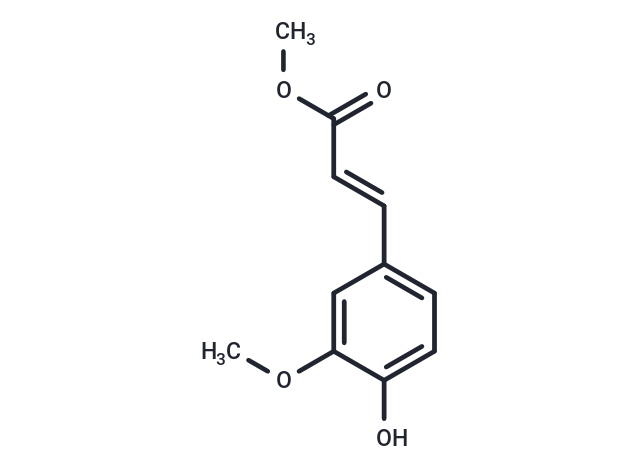
| Smiles | COC(=O)\C=C\c1ccc(O)c(OC)c1 |
| Relative Density. | 1.204 g/cm3 |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. | ||||||||||||||||||||||||||||||||||||||||
| Solubility Information | DMSO: 250 mg/mL (1200.71 mM), Sonication is recommended. Ethanol: 20 mg/mL (96.06 mM), Sonication is recommended. | ||||||||||||||||||||||||||||||||||||||||
| In Vivo Formulation | 10% DMSO+40% PEG300+5% Tween 80+45% Saline: 2 mg/mL (9.61 mM), Sonication is recommended. Please add the solvents sequentially, clarifying the solution as much as possible before adding the next one. Dissolve by heating and/or sonication if necessary. Working solution is recommended to be prepared and used immediately. The formulation provided above is for reference purposes only. In vivo formulations may vary and should be modified based on specific experimental conditions. | ||||||||||||||||||||||||||||||||||||||||
Solution Preparation Table | |||||||||||||||||||||||||||||||||||||||||
Ethanol/DMSO
DMSO
Note : The dilution table applies only to solid products. For liquid products, please calculate the stock solution based on the stated concentration and/or density. | |||||||||||||||||||||||||||||||||||||||||
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 μL PEG300
stock solution to 400 μL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 μL Tween 80 and mix well until fully clarified.
3) Add 450 μL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.