 Your shopping cart is currently empty
Your shopping cart is currently empty

Synonyms: L-(-)-Malic acid, (S)-2-Hydroxysuccinic acid, (S)-(-)-HYDROXYSUCCINIC ACID
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 25 g | $40 | - | In Stock | |
| 1 mL x 10 mM (in DMSO) | $29 | In Stock | In Stock |
| Description | (S)-Malic acid ((S)-2-Hydroxysuccinic acid) is a tart-tasting organic dicarboxylic acid found in many sour foods, such as apples, and contributes to the sourness of green apples and tartness of wine, although its concentration decreases with fruit ripeness (wikipedia). In its ionized form, it is called malate, an intermediate in the TCA cycle alongside fumarate, and can be formed from pyruvate through anaplerotic reactions. In humans, malic acid is derived from food sources and synthesized in the body via the citric acid cycle in mitochondria, playing a crucial role in energy production under both aerobic and anaerobic conditions. Under aerobic conditions, malate is oxidized to oxaloacetate, providing reducing equivalents via the malate-aspartate redox shuttle, while during anaerobic conditions, its simultaneous reduction to succinate and oxidation to oxaloacetate removes excess reducing equivalents, reversing hypoxia's inhibition of glycolysis and energy production. Studies on rats have shown that tissue malate depletes following exhaustive physical activity, suggesting that malic acid deficiency may cause physical exhaustion. Administering malic acid to rats has been shown to elevate mitochondrial malate, increasing mitochondrial respiration and energy production. |
| In vitro | It is shown that ME is essential for (S)-Malic acid (L-malic acid) utilization in L. casei. Moreover, deletion of either the gene encoding the histidine kinase or the response regulator of the TC system resulted in the loss of the ability to grow on (S)-Malic acid, thus indicating that the cognate TC system regulates and is essential for the expression of ME. Transcriptional analyses shows that expression of maeE is induced in the presence of (S)-Malic acid and repressed by glucose, whereas TC system expression is induced by (S)-Malic acid and is not repressed by glucose. |
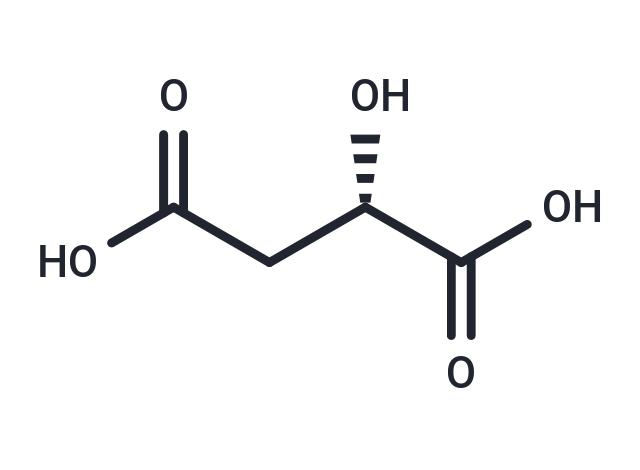
| Synonyms | L-(-)-Malic acid, (S)-2-Hydroxysuccinic acid, (S)-(-)-HYDROXYSUCCINIC ACID |
| Molecular Weight | 134.09 |
| Formula | C4H6O5 |
| Cas No. | 97-67-6 |
| Smiles | [C@H](CC(O)=O)(C(O)=O)O |
| Relative Density. | 1.60 g/cm3 |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. | |||||||||||||||||||||||||||||||||||
| Solubility Information | DMSO: 123.75 mg/mL (922.89 mM), Sonication is recommended. | |||||||||||||||||||||||||||||||||||
| In Vivo Formulation | 10% DMSO+40% PEG300+5% Tween 80+45% Saline: 2 mg/mL (14.92 mM), Sonication is recommended. 10% DMSO+90% Saline: 10 mg/mL (74.58 mM), Solution. Please add the solvents sequentially, clarifying the solution as much as possible before adding the next one. Dissolve by heating and/or sonication if necessary. Working solution is recommended to be prepared and used immediately. The formulation provided above is for reference purposes only. In vivo formulations may vary and should be modified based on specific experimental conditions. | |||||||||||||||||||||||||||||||||||
Solution Preparation Table | ||||||||||||||||||||||||||||||||||||
DMSO
Note : The dilution table applies only to solid products. For liquid products, please calculate the stock solution based on the stated concentration and/or density. | ||||||||||||||||||||||||||||||||||||
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 µL PEG300
stock solution to 400 µL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 µL Tween 80 and mix well until fully clarified.
3) Add 450 µL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.