 Your shopping cart is currently empty
Your shopping cart is currently empty

Synonyms: NSC 115829, CB1954, 5-Aziridino-2,4-dinitrobenzamide

| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 5 mg | $47 | In Stock | In Stock | |
| 10 mg | $72 | In Stock | In Stock | |
| 25 mg | $147 | In Stock | In Stock | |
| 50 mg | $222 | In Stock | In Stock | |
| 100 mg | $328 | - | In Stock | |
| 1 mL x 10 mM (in DMSO) | $52 | In Stock | In Stock |
| Description | CB1954(Tretazicar (NSC-115829)), an anticancer prodrug, is activated by NAD(P)H quinone oxidoreductase 2. It is converted in the presence of the enzyme NQO2 and co-substrate caricotamide ( EP-0152R) (EP) into a potent cytotoxic bifunctional alkylating agent. |
| In vitro | In the NPC cell line CNE1, toxic Tretazicar enhances cells killing. The overexpression of nitroreductase oxidored nitro domain-containing protein 1 (NOR1) reduce the 4 nitro group of Tretazicar, a potent cytotoxin, in order to convert the monofunctional alkylating agent Tretazicar into a toxic form. In the HepG2 cell line, the NOR1 gene upregulates of Grb2 expression and activates of MAPK signal transduction leading to enhances Tretazicar mediated cell cytotoxicity. |
| In vivo | The NTR/CB1954 system, which is in a dose-dependent effect, are used for specific ablation of cells in vivo. NTR-mediated cell killing by CB1954, which is activated cross-links, presumed through triggers the apoptosis cascade resulting in rapid cell death. Selective and potent cells killing by NTR-CB1954 does not require a functional p53. |
| Synonyms | NSC 115829, CB1954, 5-Aziridino-2,4-dinitrobenzamide |
| Cell Research | HepG2 cells,which are maintained in RPMI 1640 supplemented with 10% fetal calf serum (FCS) in a humidified culture incubator at 37?C with 5% CO2 and 95% air, grow to ~80% confluence are washed with PBS and treated with r CB1954(4-10 μmol/L) for 48hours. |
| Animal Research | RED 40 female mice,which express high levels of BLG-NTR transgene in the mammary gland and nontransgenic control mice on lactation day 6, were injected intraperitoneally (i.p.)with 50 mg/kg CB1954 dissolved in arachis oil containing 10% acetone. |
| Molecular Weight | 252.18 |
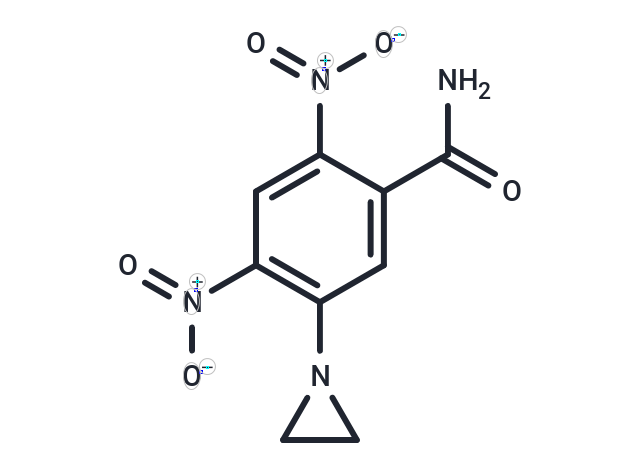
| Formula | C9H8N4O5 |
| Cas No. | 21919-05-1 |
| Smiles | NC(=O)c1cc(N2CC2)c(cc1[N+]([O-])=O)[N+]([O-])=O |
| Relative Density. | 1.671g/cm3 |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. | |||||||||||||||||||||||||||||||||||
| Solubility Information | Ethanol: < 1 mg/mL (insoluble or slightly soluble) H2O: < 1 mg/mL (insoluble or slightly soluble) DMSO: 47 mg/mL (186.37 mM), Sonication is recommended. | |||||||||||||||||||||||||||||||||||
| In Vivo Formulation | 10% DMSO+40% PEG300+5% Tween 80+45% Saline: 2 mg/mL (7.93 mM), Sonication is recommended. Please add the solvents sequentially, clarifying the solution as much as possible before adding the next one. Dissolve by heating and/or sonication if necessary. Working solution is recommended to be prepared and used immediately. The formulation provided above is for reference purposes only. In vivo formulations may vary and should be modified based on specific experimental conditions. | |||||||||||||||||||||||||||||||||||
Solution Preparation Table | ||||||||||||||||||||||||||||||||||||
DMSO
Note : The dilution table applies only to solid products. For liquid products, please calculate the stock solution based on the stated concentration and/or density. | ||||||||||||||||||||||||||||||||||||
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 µL PEG300
stock solution to 400 µL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 µL Tween 80 and mix well until fully clarified.
3) Add 450 µL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.