 Your shopping cart is currently empty
Your shopping cart is currently empty

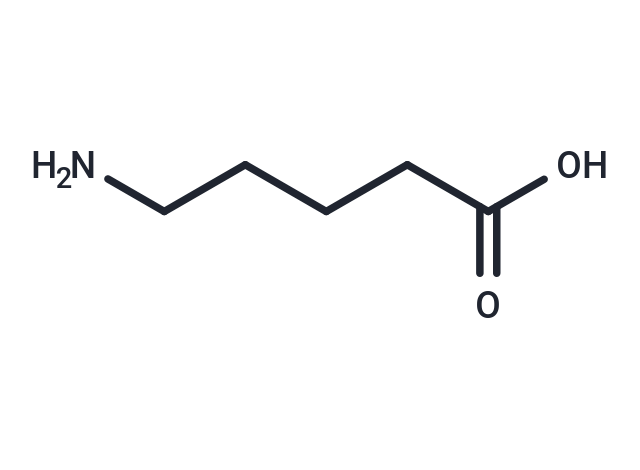
5-aminovalerate (or 5-aminopentanoic acid) is a lysine degradation product. It can be produced both endogenously or through bacterial catabolism of lysine. 5-aminovalerate is formed via the following multi-step reaction: L-lysine leads to cadverine leads to L-piperideine leads 5-aminovalerate . In other words it is a metabolite of cadaverine which is formed via the intermediate, 1-piperideine. Cadaverine is a foul-smelling diamine compound produced by protein hydrolysis during putrefaction of animal tissue. High levels of 5-aminovalerate in biofluids may indicate bacterial overgrowth or endogenous tissue necrosis. In most cases endogenous 5-aminovalerate is thought to be primarily a microbial metabolite produced by the gut or oral microflora, although it can be produced endogenously. 5-aminopentanoic acid is an in vivo substrate of 4-aminobutyrate:2-oxoglutarate aminotransferase .
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 1 g | $29 | - | In Stock | |
| 5 g | $48 | - | In Stock | |
| 10 g | $78 | - | In Stock |
| Description | 5-aminovalerate (or 5-aminopentanoic acid) is a lysine degradation product. It can be produced both endogenously or through bacterial catabolism of lysine. 5-aminovalerate is formed via the following multi-step reaction: L-lysine leads to cadverine leads to L-piperideine leads 5-aminovalerate . In other words it is a metabolite of cadaverine which is formed via the intermediate, 1-piperideine. Cadaverine is a foul-smelling diamine compound produced by protein hydrolysis during putrefaction of animal tissue. High levels of 5-aminovalerate in biofluids may indicate bacterial overgrowth or endogenous tissue necrosis. In most cases endogenous 5-aminovalerate is thought to be primarily a microbial metabolite produced by the gut or oral microflora, although it can be produced endogenously. 5-aminopentanoic acid is an in vivo substrate of 4-aminobutyrate:2-oxoglutarate aminotransferase . |
| In vitro | 5-aminovalerate is a normal metabolite present in human saliva, with a tendency to elevated concentration in patients with chronic periodontitis. Bacterial contamination and decomposition of salivary proteins is primarily responsible for elevated salivary levels. Beyond being a general waste product, 5-aminovalerate is also believed to act as a methylene homologue of gamma-aminobutyric acid (GABA) and functions as a weak GABA agonist. It is also known as an antifibrinolytic amino acid analog and so it functions as a weak inhibitor of the blood clotting pathway. |
| Molecular Weight | 117.15 |
| Formula | C5H11NO2 |
| Cas No. | 660-88-8 |
| Smiles | NCCCCC(O)=O |
| Relative Density. | 1.2000 g/cm3 (Estimated) |
| Storage | Pure form: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. | |||||||||||||||||||||||||||||||||||
| Solubility Information | DMSO: Slightly soluble H2O: 250 mg/mL (2134.02 mM) | |||||||||||||||||||||||||||||||||||
Solution Preparation Table | ||||||||||||||||||||||||||||||||||||
H2O
Note : The dilution table applies only to solid products. For liquid products, please calculate the stock solution based on the stated concentration and/or density. | ||||||||||||||||||||||||||||||||||||
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 μL PEG300
stock solution to 400 μL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 μL Tween 80 and mix well until fully clarified.
3) Add 450 μL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.