Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
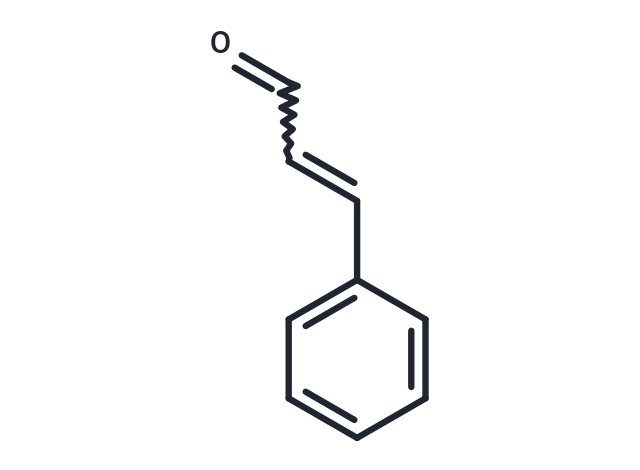
Synonyms: Cinnamic Aldehyde
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 100 mg | $30 | In Stock | In Stock | |
| 500 mg | $64 | In Stock | In Stock | |
| 1 g | $93 | - | In Stock | |
| 1 mL x 10 mM (in DMSO) | $39 | In Stock | In Stock |
| Description | 1. Cinnamaldehyde (Cinnamic Aldehyde) has antipyretic activity. 2. Cinnamaldehyde is a sedative agent. 3. Cinnamaldehyde inhibits invasive capabilities of MDA-MB-435S cells was correlated with down-regulating the expression of miR-27a. 4. Cinnamaldehyde induces the generation of reactive oxygen species and exerts vasodilator and anticancer effects. 5. Cinnamaldehyde appears to be a promising candidate as an adjuvant in combination therapy with 5-fluorouracil (5-FU) and oxaliplatin (OXA), two chemotherapeutic agents used in CRC treatment. The possible mechanisms of its action may involve the regulation of drugmetabolizing genes. 6. Cinnamaldehyde plays a certain role in inhibiting the occurrence and progression of melanoma and its action mechanism may be manifested by inhibiting expression of VEGF and HIF-α, thus blood vessel simulation and formation of new blood vessels of melanoma cells, and growth of tumors accordingly. |
| Synonyms | Cinnamic Aldehyde |
| Molecular Weight | 132.16 |
| Formula | C9H8O |
| Cas No. | 104-55-2 |
| Smiles | O=CC=CC=1C=CC=CC1 |
| Relative Density. | 1.05 g/cm3 at 25℃ (lit.) |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. | |||||||||||||||||||||||||||||||||||
| Solubility Information | DMSO: 255 mg/mL (1929.48 mM), Sonication is recommended. | |||||||||||||||||||||||||||||||||||
Solution Preparation Table | ||||||||||||||||||||||||||||||||||||
DMSO
Note : The dilution table applies only to solid products. For liquid products, please calculate the stock solution based on the stated concentration and/or density. | ||||||||||||||||||||||||||||||||||||
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 μL PEG300
stock solution to 400 μL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 μL Tween 80 and mix well until fully clarified.
3) Add 450 μL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.