Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
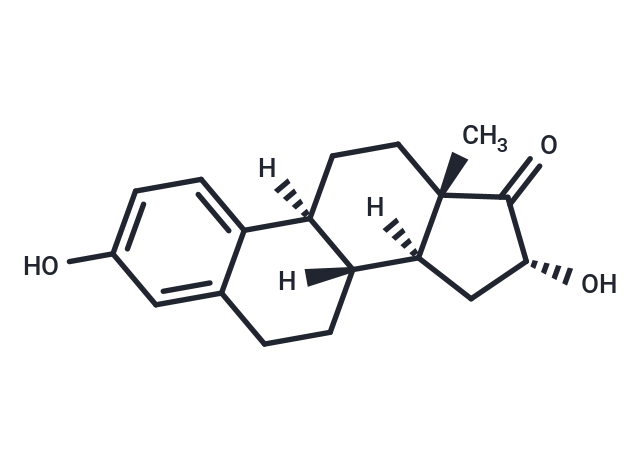
Synonyms: 16αOHE
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 1 mg | $98 | - | In Stock | |
| 5 mg | $297 | - | In Stock | |
| 10 mg | $479 | - | In Stock | |
| 25 mg | $959 | - | In Stock | |
| 50 mg | $1,530 | - | In Stock | |
| 1 mL x 10 mM (in DMSO) | $327 | - | In Stock |
| Description | 16α-Hydroxyestrone is an important metabolite of estradiol, with important application value in basic research of metabolic diseases. It acts as an inhibitor of Breast cancer cell line MDA-MB-175-VII. |
| In vitro | Methods: The contents of two active estrogen metabolites of 16α-Hydroxyestrone in the body were detected, and their correlation with bone mineral density was analyzed. Results: The upregulation of active 16α-Hydroxyestrone and E3 levels was positively correlated with increased bone mineral density [1]. |
| Synonyms | 16αOHE |
| Molecular Weight | 286.37 |
| Formula | C18H22O3 |
| Cas No. | 566-76-7 |
| Smiles | C[C@@]12[C@]([C@]3([C@@](C=4C(CC3)=CC(O)=CC4)(CC1)[H])[H])(C[C@@H](O)C2=O)[H] |
| Relative Density. | 1.249 g/cm3 (Predicted) |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. |
| Solubility Information | DMSO: 200 mg/mL (698.4 mM), Sonication is recommended. DMF: 30 mg/mL (104.76 mM), Sonication is recommended. DMF:PBS (pH 7.2) (1:5): 0.15 mg/mL (0.52 mM), Sonication is recommended. |
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 µL PEG300
stock solution to 400 µL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 µL Tween 80 and mix well until fully clarified.
3) Add 450 µL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.