 Your shopping cart is currently empty
Your shopping cart is currently empty

N-Acetyl-L-cysteine ethyl ester (NACET) is a derivative of the amino acid L-cysteine. NACET has been shown to exhibit diverse biochemical and physiological effects, including inhibition of protein synthesis, inhibition of DNA replication, and induction of apoptosis. Additionally, it has demonstrated anti-inflammatory, antioxidant, and anti-cancer activity.
| Pack Size | Price | USA Warehouse | Global Warehouse | Quantity |
|---|---|---|---|---|
| 5 mg | $30 | - | In Stock | |
| 10 mg | $40 | - | In Stock | |
| 25 mg | $71 | - | In Stock | |
| 50 mg | $113 | - | In Stock | |
| 100 mg | $198 | - | In Stock | |
| 200 mg | $298 | - | In Stock | |
| 1 mL x 10 mM (in DMSO) | $44 | - | In Stock |
| Description | N-Acetyl-L-cysteine ethyl ester (NACET) is a derivative of the amino acid L-cysteine. NACET has been shown to exhibit diverse biochemical and physiological effects, including inhibition of protein synthesis, inhibition of DNA replication, and induction of apoptosis. Additionally, it has demonstrated anti-inflammatory, antioxidant, and anti-cancer activity. |
| In vitro | Under concomitant oxidative stress induced by 2 mM H2O2, N-Acetyl-L-cysteine ethyl ester (NACET) showed strong and significant protective effect on RPE cells when the concentration was 0.4 mM. Moreover, NACET treatment also resulted in an enhanced protective effect when RPE cells were stressed by the organic ROS generator peroxide tert-Butyl hydroperoxide (t-BOOH).NACET significantly increased both intracellular GSH and cysteine at 0.2 and 1 mM concentrations, respectively, as well as extracellular total GSH[2]. |
| Synonyms | N-Acetylcysteine ethyl ester, NACET |
| Molecular Weight | 191.25 |
| Formula | C7H13NO3S |
| Cas No. | 59587-09-6 |
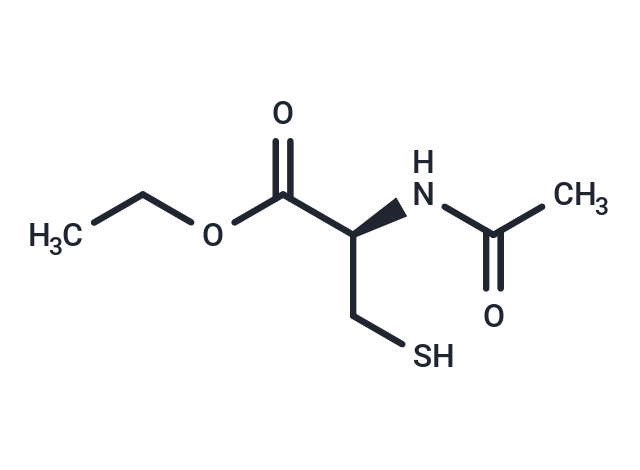
| Smiles | CCOC(=O)[C@H](CS)NC(C)=O |
| Relative Density. | 1.138 g/cm3 (Predicted) |
| Storage | store at low temperature | Powder: -20°C for 3 years | In solvent: -80°C for 1 year | Shipping with blue ice/Shipping at ambient temperature. | |||||||||||||||||||||||||||||||||||
| Solubility Information | DMSO: 45 mg/mL (235.29 mM), Sonication is recommended. H2O: 90 mg/mL (470.59 mM), Sonication is recommended. | |||||||||||||||||||||||||||||||||||
| In Vivo Formulation | 10% DMSO+40% PEG300+5% Tween 80+45% Saline: 1 mg/mL (5.23 mM), Sonication is recommended. Please add the solvents sequentially, clarifying the solution as much as possible before adding the next one. Dissolve by heating and/or sonication if necessary. Working solution is recommended to be prepared and used immediately. The formulation provided above is for reference purposes only. In vivo formulations may vary and should be modified based on specific experimental conditions. | |||||||||||||||||||||||||||||||||||
Solution Preparation Table | ||||||||||||||||||||||||||||||||||||
DMSO/H2O
| ||||||||||||||||||||||||||||||||||||
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 μL PEG300
stock solution to 400 μL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 μL Tween 80 and mix well until fully clarified.
3) Add 450 μL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.