Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
Synonyms:
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 25 mg | $1,970 | 8-10 weeks | 8-10 weeks | |
| 50 mg | $2,580 | 8-10 weeks | 8-10 weeks | |
| 100 mg | $3,400 | 8-10 weeks | 8-10 weeks |
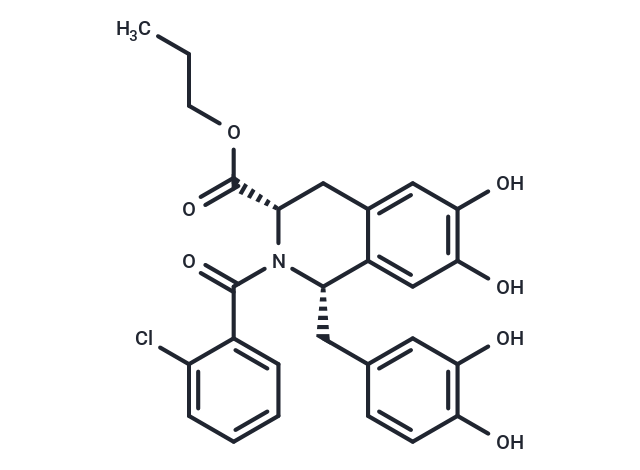
| Description | Influenza virus-IN-6 (Compound 35) is a potent inhibitor of the N-terminal domain of the polymerase acidic protein (PA N) endonuclease subunit of the influenza virus, with an inhibitory concentration 50 (IC 50) value of 0.20 μM [1]. |
| In vitro | Influenza virus-IN-6 (Compound 35) exhibits anti-influenza virus activity with EC50 values of 1.28 ± 0.35, 1.12 ± 0.65, 0.76 ± 0.11, and 0.43 ± 0.06 μM against H1N1, H5N1, H3N2, and Flu B, respectively, in MDCK cells after 48 hours [1]. At 5-20 μM for 24 hours, it affects viral replication without impacting viral particles, cellular adsorption, or release [1]. Additionally, at 2.5-10 μM for 24 hours, Influenza virus-IN-6 inhibits influenza virus polymerase activity [1]. The compound also demonstrates good stability in mouse plasma, liver microsomes, and intestinal S9-UDPGA [1]. |
| In vivo | Influenza virus-IN-6 (Compound 35) administered intraperitoneally at 7.5-30 mg/kg/d twice daily for seven days significantly protected mice against influenza virus infection [1]. In rats (n = 5), the pharmacokinetic profile of Influenza virus-IN-6 (Compound 35) after a single dose revealed the following parameters: for intravenous (IV) administration (2 mg/kg), half-life (T 1/2) was 0.33 ± 0.07 hours, peak plasma concentration (C max) was 1586.55 ± 366.48 ng/mL, area under the curve (AUC 0-t) was 536.45 ± 58.72 h·ng/mL, and clearance (CL) was 53.76 ± 13.18 mL/min/kg; for oral (PO) administration (10 mg/kg), T 1/2 was 0.82 ± 0.16 hours, time to maximum concentration (T max) was 0.52 hours, C max was 92.20 ± 36.25 ng/mL, AUC 0-t was 164.30 ± 26.37 h·ng/mL, and bioavailability (F %) was 6.13%; for intraperitoneal (IP) injection (15 mg/kg), T 1/2 was 1.07 ± 0.25 hours, T max was 0.45 hours, C max was 889.52 ± 233.17 ng/mL, AUC 0-t was 790.62 ± 188.31 h·ng/mL, and F % was 29.50% [1]. IV represents intravenous injection, IP represents intraperitoneal injection, and PO represents the gastrointestinal route. |
| Molecular Weight | 511.95 |
| Formula | C27H26ClNO7 |
| Cas No. | 2919303-26-5 |
| Smiles | CCCOC(=O)[C@@H]1CC2=CC(=C(C=C2[C@@H](N1C(=O)C3=CC=CC=C3Cl)CC4=CC(=C(C=C4)O)O)O)O |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. |
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 µL PEG300
stock solution to 400 µL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 µL Tween 80 and mix well until fully clarified.
3) Add 450 µL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.