Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
Synonyms:
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 10 mg | Inquiry | 10-14 weeks | 10-14 weeks | |
| 50 mg | Inquiry | 10-14 weeks | 10-14 weeks |
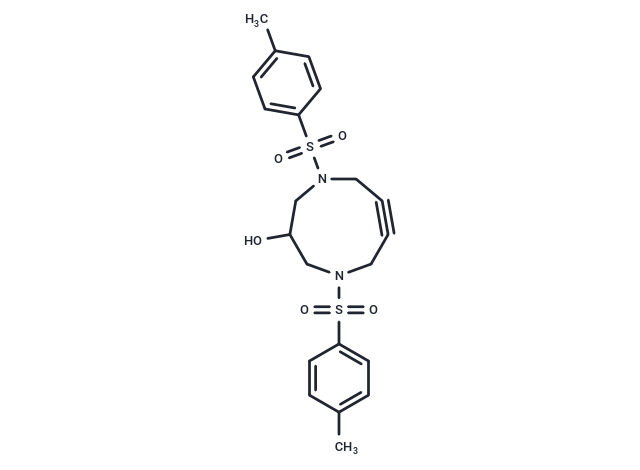
| Description | DACN(Tos2,6-OH) serves as a potent click chemistry reagent featuring an Azide group and a distinctively bent alkyne moiety. This structure enhances its reactivity for cycloaddition reactions compared to nonbent acyclic alkynes, especially evident in cyclononyne alkynes which exhibit substantially higher reactivity. The bent configuration adds both electronic and steric effects that intensify the reactivity, facilitating strain-promoted azide-alkyne cycloaddition (SPAAC). SPAAC with cycloalkynes is commonly utilized for dependable molecular conjugation across various fields. Furthermore, DACN(Tos2,6-OH) can participate in copper-catalyzed azide-alkyne cycloaddition (CuAAc), coupling efficiently with molecules that contain Azide groups. Compared to cyclooctynes, DACNs demonstrate superior thermal and chemical stabilities while maintaining similar click reactivity [1]. Nitrogens within the compound provide versatile linking sites for numerous functional units. |
| Molecular Weight | 448.56 |
| Formula | C21H24N2O5S2 |
| Cas No. | 2109751-74-6 |
| Smiles | S(=O)(=O)(C1=CC=C(C)C=C1)N2CC(O)CN(S(=O)(=O)C3=CC=C(C)C=C3)CC#CC2 |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. |
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 µL PEG300
stock solution to 400 µL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 µL Tween 80 and mix well until fully clarified.
3) Add 450 µL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.