 Your shopping cart is currently empty
Your shopping cart is currently empty

Synonyms:
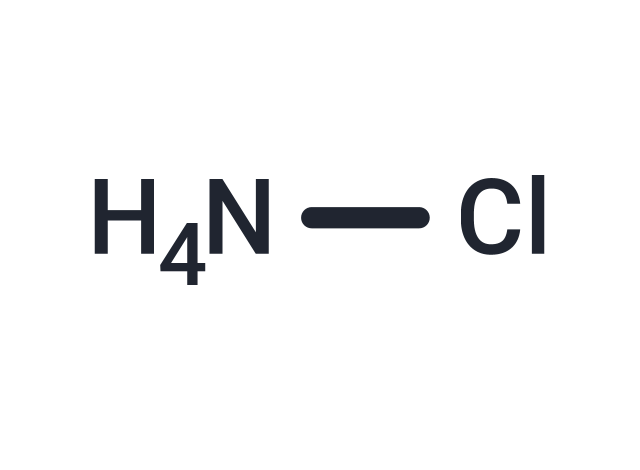

| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 200 mg | $35 | In Stock | In Stock | |
| 500 mg | $55 | In Stock | In Stock | |
| 1 g | $80 | In Stock | In Stock | |
| 5 g | $139 | - | In Stock | |
| 10 g | $172 | - | In Stock | |
| 25 g | $288 | - | In Stock |
| Description | Ammonium chloride is a compound commonly used in molecular biology research. As a dipolar compound that regulates pH, it can induce intracellular alkalinization and metabolic acidosis, thereby affecting enzyme activity and biological processes. Additionally, ammonium chloride acts as an inhibitor of autophagy and lysosomal function, and can be used to establish kidney stone models. |
| In vitro | Ammonium Chloride (NH4Cl) reduces the yield of reovirus during infection of mouse L cells.[2] |
| In vivo | Ammonium Chloride (0.28 M in drinking water; 8-9-week-old C57B/L6 mice) promotes the survival of myocardial cells in vivo by decreasing contractile dysfunction, cardiac hypertrophy, inflammation, apoptosis and autophagy. Ammonium Chloride effectively improved doxorubicin (DOX)-induced cardiomyocyte apoptosis and cardiac dysfunction in mice.[1] |
| Disease Modeling Protocol | Kidney stone model
*Precautions: After euthanizing the animal on day 28, the kidneys were quickly separated. |
| Molecular Weight | 53.49 |
| Formula | ClH4N |
| Cas No. | 12125-02-9 |
| Smiles | Cl[NH4] |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. | |||||||||||||||||||||||||||||||||||
| Solubility Information | DMSO: 52 mg/mL (972.14 mM), Sonication is recommended. H2O: 80 mg/mL (1495.61 mM), Sonication is recommended. | |||||||||||||||||||||||||||||||||||
| In Vivo Formulation | 10% DMSO+40% PEG300+5% Tween-80+45% Saline: 2.5 mg/mL (46.74 mM), Sonication is recommended. Please add the solvents sequentially, clarifying the solution as much as possible before adding the next one. Dissolve by heating and/or sonication if necessary. Working solution is recommended to be prepared and used immediately. The formulation provided above is for reference purposes only. In vivo formulations may vary and should be modified based on specific experimental conditions. | |||||||||||||||||||||||||||||||||||
Solution Preparation Table | ||||||||||||||||||||||||||||||||||||
DMSO/H2O
Note : The dilution table applies only to solid products. For liquid products, please calculate the stock solution based on the stated concentration and/or density. | ||||||||||||||||||||||||||||||||||||
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 µL PEG300
stock solution to 400 µL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 µL Tween 80 and mix well until fully clarified.
3) Add 450 µL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.