 Your shopping cart is currently empty
Your shopping cart is currently empty

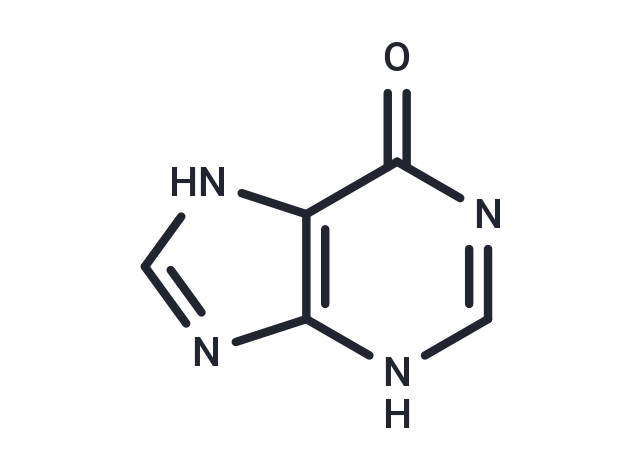
Synonyms: Sarkin, Sarcine, Purin-6-ol, 6-Hydroxypurine
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 25 mg | $30 | In Stock | In Stock | |
| 50 mg | $37 | In Stock | In Stock | |
| 100 mg | $50 | In Stock | In Stock | |
| 500 mg | $113 | In Stock | In Stock | |
| 1 g | $163 | In Stock | In Stock | |
| 1 mL x 10 mM (in DMSO) | $45 | In Stock | In Stock |
| Description | Hypoxanthine (Purin-6-ol), also known as purine-6-ol or Hyp. Hypoxanthine is a naturally occurring purine derivative and a reaction intermediate in the metabolism of adenosine and in the formation of nucleic acids by the nucleotide salvage pathway. Under normal circumstances hypoxanthine is readily converted to uric acid.hypoxanthine is first oxidized to xanthine, which is further oxidized to uric acid by xanthine oxidase. |
| In vitro | Hypoxanthine is a naturally occurring purine derivative. It is occasionally found as a constituent of nucleic acids, where it is present in the anticodon of tRNA in the form of its nucleoside inosine. It has a tautomer known as 6-hydroxypurine. Hypoxanthine is a necessary additive in a certain cell, bacteria, and parasite cultures as a substrate and nitrogen source. [1] |
| In vivo | A linear increase of plasma hypoxanthine with duration of hypoxemia is found in pigs, and there is no difference between arterial and venous plasma. There are good correlations between hypoxanthine and lactate, base deficit and pH. Moreover, there is a direct relationship between survival time and an increase in plasma hypoxanthine. Survival time correlates negatively with the rate of hypoxanthine increase (r=-0.62). All animals die when hypoxanthine exceeds 125 pM/liter. The increase of hypoxanthine, thus, reflected the prognosis of acute hypoxia in contrast to base deficit[1]. |
| Synonyms | Sarkin, Sarcine, Purin-6-ol, 6-Hydroxypurine |
| Molecular Weight | 136.11 |
| Formula | C5H4N4O |
| Cas No. | 68-94-0 |
| Smiles | O=C1N=CNC=2N=CNC12 |
| Relative Density. | 1.4295 g/cm3 (Estimated) |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. | |||||||||||||||||||||||||
| Solubility Information | Ethanol: Insoluble DMSO: 5 mg/mL (36.73 mM), Sonication is recommended. | |||||||||||||||||||||||||
Solution Preparation Table | ||||||||||||||||||||||||||
DMSO
Note : The dilution table applies only to solid products. For liquid products, please calculate the stock solution based on the stated concentration and/or density. | ||||||||||||||||||||||||||
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 µL PEG300
stock solution to 400 µL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 µL Tween 80 and mix well until fully clarified.
3) Add 450 µL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.