Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
Synonyms: Tumor Necrosis Factor Ligand Superfamily Member 2, Tumor Necrosis Factor, TNF-α, TNFSF2, TNF-Alpha, TNF-a, TNFA, TNF α, TNF, Cachectin

| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 5 µg | $35 | - | In Stock | |
| 10 µg | $53 | - | In Stock | |
| 20 µg | $77 | - | In Stock | |
| 50 µg | $129 | 7-10 days | 7-10 days | |
| 100 µg | $215 | 7-10 days | 7-10 days | |
| 200 µg | $363 | 7-10 days | 7-10 days | |
| 500 µg | $739 | 7-10 days | 7-10 days | |
| 1 mg | $1,060 | 7-10 days | 7-10 days |
| Bioactivity | Measured in a cytotoxicity assay using L‑929 mouse fibroblast cells in the presence of the metabolic inhibitor actinomycin D. The ED50 for this effect is 10-40 pg/ml. (QC verified) |
| Description | TNF alpha Protein, Human, Recombinant (aa 77-233, His) is expressed in E. coli expression system with C-6xHis tag. The predicted molecular weight is 16 KDa and the accession number is P01375. |
| Species | Human |
| Expression System | E. coli |
| Tag | C-6xHis |
| Accession Number | P01375 |
| Amino Acid | Val77-Leu233 |
| Construction | Val77-Leu233 |
| Protein Purity | Greater than 95% as determined by reducing SDS-PAGE. (QC verified) 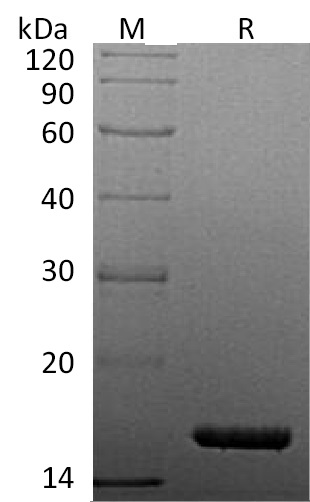 |
| Endotoxin | < 0.1 ng/µg (1 EU/µg) as determined by LAL test. |
| Formulation | Lyophilized from a solution filtered through a 0.22 μm filter, containing 20 mM Tris-HCl, 150 mM NaCl, pH 8.0. |
| Reconstitution | Reconstitute the lyophilized protein in distilled water. The product concentration should not be less than 100 μg/ml. Before opening, centrifuge the tube to collect powder at the bottom. After adding the reconstitution buffer, avoid vortexing or pipetting for mixing. |
| Synonyms | Tumor Necrosis Factor Ligand Superfamily Member 2, Tumor Necrosis Factor, TNF-α, TNFSF2, TNF-Alpha, TNF-a, TNFA, TNF α, TNF, Cachectin |
| Research Background | Tumor Necrosis Factor-α (TNF-α) is secreted by macrophages, monocytes, neutrophils, T-cells, and NK-cells following stimulation by bacterial LPS. Cells expressing CD4 secrete TNF-α while cells that express CD8 secrete little or no TNF-α. Synthesis of TNF-α can be induced by many different stimuli including interferons, IL2, and GM-CSF. The clinical use of the potent anti-tumor activity of TNF-α has been limited by the proinflammatory side effects such as fever, dose-limiting hypotension, hepatotoxicity, intravascular thrombosis, and hemorrhage. Designing clinically applicable TNF-α mutants with low systemic toxicity has been of intense pharmacological interest. Human TNF-α that binds to murine TNF-R55 but not murine TNF-R7, exhibits retained anti-tumor activity and reduced systemic toxicity in mice compared with murine TNF-α, which binds to both murine TNF receptors. Based on these results, many TNF-α mutants that selectively bind to TNF-R55 have been designed. These mutants displayed cytotoxic activities on tumor cell lines in vitro and have exhibited lower systemic toxicity in vivo. Recombinant Human TNF-α High Active Mutant differs from the wild-type by amino acid subsitution of amino acids 1-7 with Arg8, Lys9, Arg10 and Phe157. This mutant form has been shown to have increased activity with less inflammatory side effects in vivo. |
| Molecular Weight | 16 KDa (reducing condition) |
| Shipping | In general, lyophilized powders are shipped with blue ice, while solutions are shipped with dry ice. |
| Storage | Lyophilized powders can be stably stored for over 12 months, while liquid products can be stored for 6-12 months at -80°C. For reconstituted protein solutions, the solution can be stored at -20°C to -80°C for at least 3 months. Please avoid multiple freeze-thaw cycles and store products in aliquots. |
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.