Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
Synonyms:
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 25 mg | $1,520 | 1-2 weeks | 1-2 weeks | |
| 50 mg | $1,980 | 1-2 weeks | 1-2 weeks | |
| 100 mg | $2,500 | 1-2 weeks | 1-2 weeks |
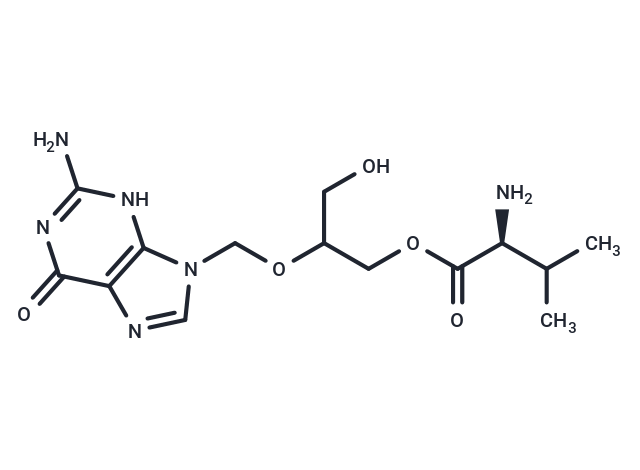
| Description | Valganciclovir, the L-valyl ester of ganciclovir, is a prodrug effectively converted to ganciclovir in the body, serving as an antiviral medication for cytomegalovirus (CMV) infections. In vitro studies demonstrate that valganciclovir inhibits glycylsarcosine transport mediated by PEPT1 and PEPT2 in Caco-2 and SKPT cells, respectively, with competitive inhibition K(i) values of 1.68±0.30 mM for PEPT1 and 0.043±0.005 mM for PEPT2. Clinical trials indicate valganciclovir's non-inferior efficacy to ganciclovir in pre-emptive therapy for CMV, exhibiting no significant difference in viral clearance rates or toxicity levels between the treatments. Specifically, 89.5% of patients treated with valganciclovir and 83% treated with ganciclovir achieved viral clearance at 28 days (P=0.030 for non-inferiority). Additionally, valganciclovir prophylaxis at 450 mg twice daily prevented CMV reactivation in patients on an alemtuzumab-containing regimen, with none of the 20 patients experiencing reactivation (P=0.004). |
| Molecular Weight | 354.36 |
| Formula | C14H22N6O5 |
| Cas No. | 175865-60-8 |
| Smiles | C(OC(COC([C@H](C(C)C)N)=O)CO)N1C2=C(N=C1)C(=O)N=C(N)N2 |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. |
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 µL PEG300
stock solution to 400 µL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 µL Tween 80 and mix well until fully clarified.
3) Add 450 µL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.