Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
Synonyms:
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 1 mg | $249 | - | In Stock |
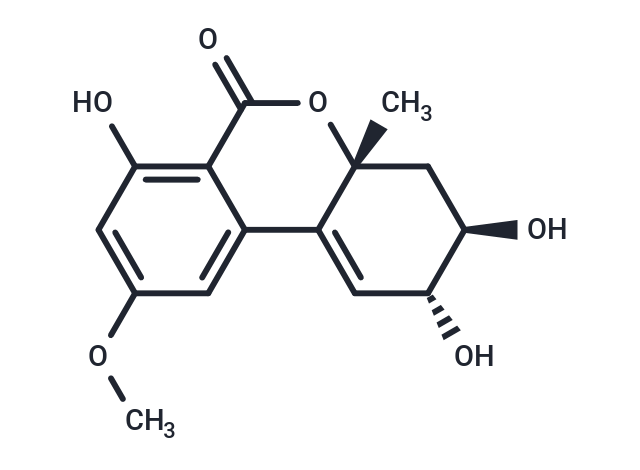
| Description | Altenuene is a mycotoxin, it frequently occurs in food and feed items infested by fungi of the genus Alternaria. Altenuene demonstrates moderate activity against Staphylococcus aureus. Altenuene also exhibits cytotoxic activity against lung cancer cell line A549, breast cancer cell line MDA-MB-231 and pancreatic cancer cell line PANC-1. |
| In vitro | METHODS AND RESULTS:Two new polyketides, 7-hydroxy-3, 5-dimethyl-isochromen-1-one (1) and 6-hydroxy-8-methoxy-3a-methyl-3a,9b-dihydro-3H-furo[3,2-c]isochromene-2,5-dione (2), along with eleven known compounds, 5'-methoxy-6-methyl-biphenyl-3,4,3'-triol (3), 7-hydroxy-3-(2-hydroxy-propyl)-5-methyl-isochromen-1-one (4), rubralactone (5), isoAltenuene (6), Altenuene (7), dihydroAltenuenes A (8), altenusin (9), alterlactone (10), 6-O-methylnorlichexanthone (11), norlichexanthone (12), and griseoxanthone C (13) were isolated from the culture of the endolichenic fungus Ulocladium sp. Compound 2 was obtained as a racemate with an unprecedented chemical skeleton. The NMR data assignments for 3 and 4 were achieved for the first time. Compounds 1-13 were screened for their antimicrobial and radical scavenging activities. CONCLUSIONS: Compound 1 showed some antifungal activity against Candida albicans SC 5314 with IC(50) of 97.93 ± 1.12 μM. Compounds 11-13 showed strong activity against Bacillus subtilis with IC(50) in the range of 1-5 μM. Compound 12 significantly inhibited the growth of methicillin-resistant Staphylococcus aureus with IC(50) of 20.95 ± 1.56 μM. Compounds 9 and 10 showed strong radical scavenging activity in comparison with vitamin C. The plausible biosynthetic pathways for compounds 1, 2, and 4-8 were discussed. |
| Molecular Weight | 292.28 |
| Formula | C15H16O6 |
| Cas No. | 29752-43-0 |
| Smiles | C[C@@](C[C@H]1O)(C(c2cc(OC)cc(O)c22)=C[C@H]1O)OC2=O |
| Relative Density. | no data available |
| Storage | Store at low temperature,Keep away from direct sunlight,Keep away from moisture Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. |
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 µL PEG300
stock solution to 400 µL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 µL Tween 80 and mix well until fully clarified.
3) Add 450 µL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.