Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
Synonyms: (-)-SNI-2011, (-)-AF102B hydrochloride hemihydrate
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 25 mg | Inquiry | 3-6 months | 3-6 months | |
| 50 mg | Inquiry | 3-6 months | 3-6 months | |
| 100 mg | Inquiry | 3-6 months | 3-6 months |
| Description | Cevimeline hydrochloride hemihydrate ((-)-SNI-2011), a novel muscarinic receptor agonist, is being explored as a potential treatment for xerostomia in Sjogren's syndrome, exhibiting an IC50 value indicative of its affinity for mAChR. This compound's pharmacological effects on the gastrointestinal, urinary, and reproductive systems, alongside its impact on various tissues, were thoroughly examined in species including mice, rats, guinea pigs, rabbits, and dogs. The metabolic breakdown of (-)-SNI-2011 was studied in vitro using rat and dog liver microsomes to assess its biotransformation. Upon oral administration, peak plasma concentrations were reached within an hour in both rats and dogs, showcasing rapid absorption and a subsequent decrease in concentration with a half-life ranging from 0.4 to 1.1 hours. Bioavailability was noted at 50% in rats and 30% in dogs. Metabolic pathways highlighted significant species differences, with both S- and N-oxidized metabolites identified in rats, but only N-oxidized metabolites in dogs. Additionally, gender differences in pharmacokinetics were observed in rats but were absent in dogs. In vitro studies pinpointed the involvement of cytochrome P450 (CYP) and flavin-containing monooxygenase (FMO) in the metabolism of (-)-SNI-2011, specifically through sulfoxidation and N-oxidation processes, respectively. CYP2D and CYP3A were identified as the primary enzymes responsible for sulfoxidation in rat liver microsomes. |
| Synonyms | (-)-SNI-2011, (-)-AF102B hydrochloride hemihydrate |
| Molecular Weight | 244.78 |
| Formula | C10H19ClNO1.5S |
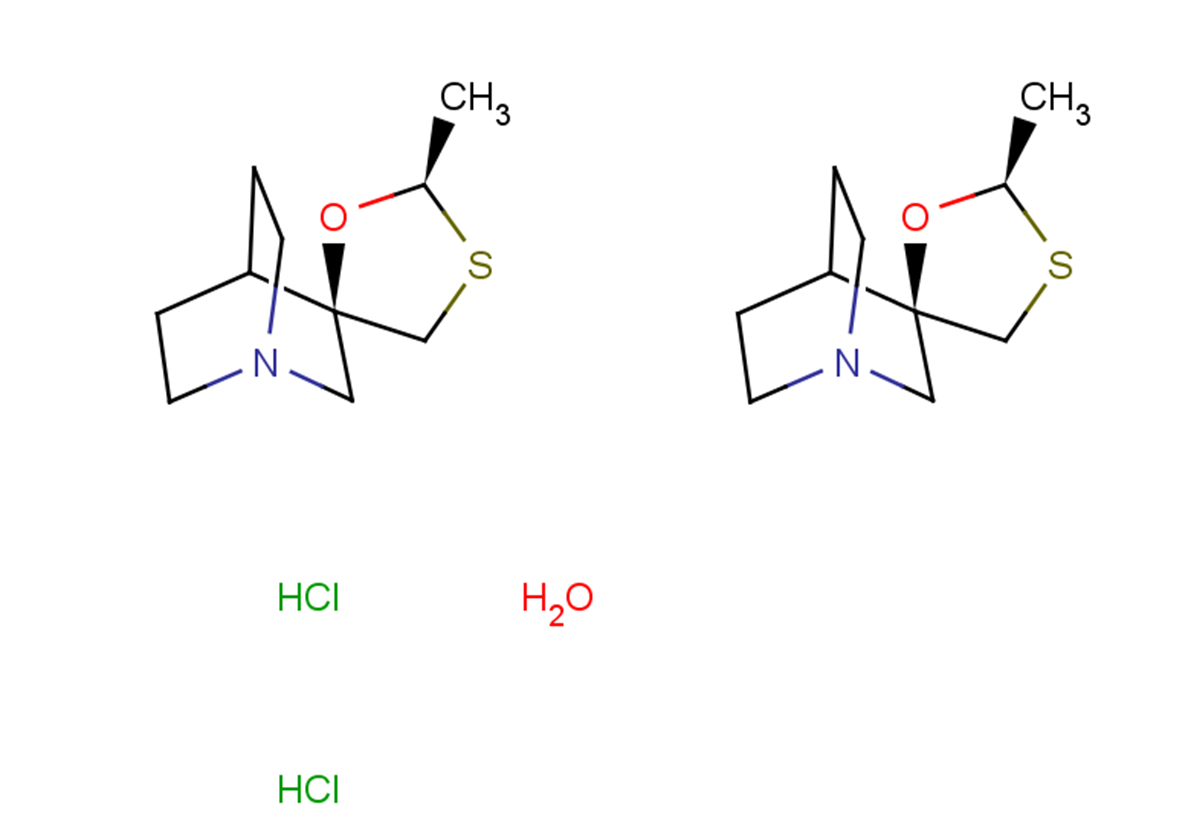
| Smiles | C[C@@H](SC1)O[C@@]21CN3CCC2CC3.Cl.[0.5H2O] |
| Relative Density. | 1.31g/cm3 |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. |
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 μL PEG300
stock solution to 400 μL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 μL Tween 80 and mix well until fully clarified.
3) Add 450 μL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.