Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
Synonyms: Solvent Green 7, HPTS
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 500 mg | $46 | - | In Stock | |
| 1 g | $79 | - | In Stock | |
| 1 mL x 10 mM (in DMSO) | $51 | In Stock | In Stock |
| Description | Pyranine (Solvent Green 7) is reported as a new class of fluorescent chemosensor for the Cu+ ion. |
| In vitro | I. Cu2+ ion detection 1. Material preparation: 1) Pyranine solution: Pyranine is usually dissolved in an appropriate solvent, such as water, PBS or other buffer, to a concentration of 1-10 μM. 2) Cu+ ion solution: Prepare Cu+ ion solutions of different concentrations. 3) Fluorescence spectrometer: A device that can provide an excitation wavelength of 450 nm and an emission wavelength of 510 nm. 2. Steps: 1) Solution preparation: Prepare Pyranine solution and prepare solutions of different concentrations containing Cu+ ions. 2) Reaction: Add Pyranine solution to the solution containing Cu+ ions and mix thoroughly. 3) Fluorescence measurement: Use a fluorescence spectrometer to measure the fluorescence intensity at an excitation wavelength of 450 nm and an emission wavelength of 510 nm. 4) Data analysis: By measuring the fluorescence changes of Cu+ ions at different concentrations, the response of Pyranine in this environment is analyzed for quantitative analysis of Cu+ ion concentration. 2. pH sensitivity analysis 1. Material preparation: 1) Pyranine solution: As mentioned above, prepare a Pyranine solution of appropriate concentration. 2) pH buffer: Prepare buffers of different pH. 2. Steps: 1) Solution preparation: Prepare Pyranine solution and prepare buffers of different pH. 2) Measure pH changes: Add Pyranine solution to buffers of different pH and record its fluorescence intensity. 3) Fluorescence analysis: Analyze the response of Pyranine to pH based on the changes in fluorescence intensity. The above information is based on published literature. Experimental procedures should be appropriately modified to meet specific research demands. |
| Synonyms | Solvent Green 7, HPTS |
| Molecular Weight | 524.39 |
| Formula | C16H7Na3O10S3 |
| Cas No. | 6358-69-6 |
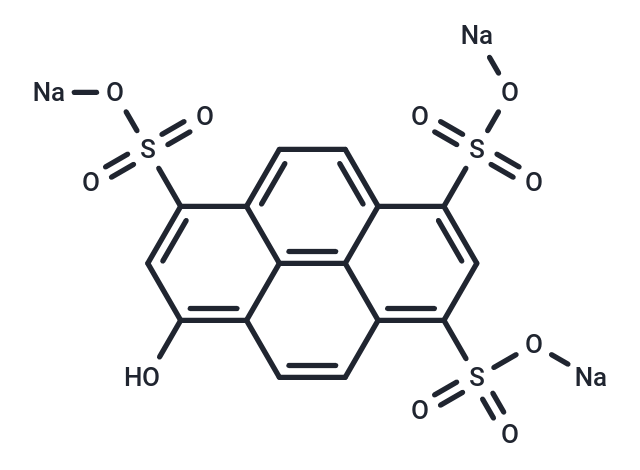
| Smiles | O=S(C1=C(C2=C34)C=CC4=C(O)C=C(S(=O)(O[Na])=O)C3=CC=C2C(S(=O)(O[Na])=O)=C1)(O[Na])=O |
| Relative Density. | 2.15 |
| Storage | Keep away from direct sunlight Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. | ||||||||||||||||||||||||||||||||||||||||
| Solubility Information | H2O: 100 mg/mL (190.7 mM), Sonication is recommended. DMSO: 6.88 mg/mL (13.12 mM), Sonication is recommended. | ||||||||||||||||||||||||||||||||||||||||
Solution Preparation Table | |||||||||||||||||||||||||||||||||||||||||
DMSO/H2O
H2O
Note : The dilution table applies only to solid products. For liquid products, please calculate the stock solution based on the stated concentration and/or density. | |||||||||||||||||||||||||||||||||||||||||
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 µL PEG300
stock solution to 400 µL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 µL Tween 80 and mix well until fully clarified.
3) Add 450 µL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.