Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
Synonyms: Quabodepistat, OPC167832, OPC 167832
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 25 mg | $1,170 | 10-14 weeks | 10-14 weeks | |
| 50 mg | $1,530 | 10-14 weeks | 10-14 weeks | |
| 100 mg | $2,440 | 10-14 weeks | 10-14 weeks |
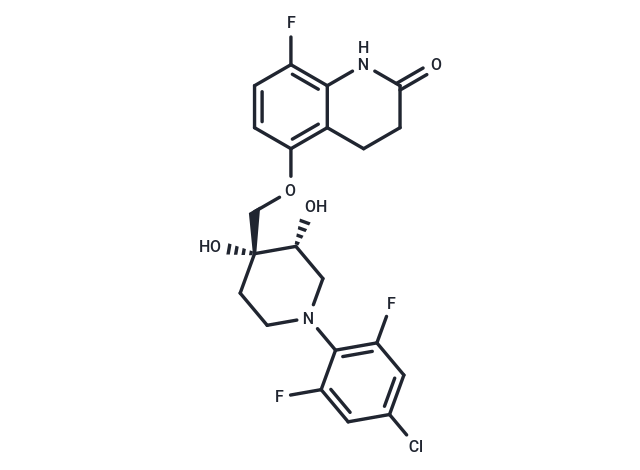
| Description | OPC-167832 is a highly potent and orally bioavailable dprE1 inhibitor with an IC₅₀ of 0.258 μM. It exhibits anti-mycobacterial activity against Mycobacterium tuberculosis and can be used in tuberculosis-related research. |
| In vitro | Methods: In vitro drug sensitivity testing of OPC-167832 was performed on Mycobacterium tuberculosis and various standard strains, and its bacteriostatic and bactericidal activities against intracellular Mycobacterium tuberculosis were determined. Results: 1 OPC-167832 exhibited potent inhibitory activity against the Mycobacterium tuberculosis laboratory strains H37Rv and Kurono, with a MIC of 0.0005 μg/mL for both, while it had almost no antibacterial activity against standard strains of aerobic and anaerobic non-mycobacterial bacteria. 2 The IC₉₀ of OPC-167832 against intracellular Mycobacterium tuberculosis was less than 100%, and the bacteriostatic concentrations against H37Rv and Kurono strains were 0.0048 μg/mL and 0.0027 μg/mL, respectively; it could exert intracellular bactericidal effect at low concentrations, and the bactericidal activity tended to be saturated when the concentration was ≥0.004 μg/mL [1]. |
| In vivo | Methods: OPC-167832 was administered orally at doses of 0.625–10 mg/kg to detect its pharmacokinetics and lung tissue distribution; oral administration at the same doses was performed for 4 weeks to observe changes in lung tissue CFU; intragastric administration of 2.5 mg/kg combined with DCMB was conducted for 12 weeks to evaluate the combined therapeutic effect. Results: 1 OPC-167832 had good pharmacokinetic properties after oral administration, with a time to peak concentration (tmax) of 0.5–1.0 h and an elimination half-life (t1/2) of 1.3–2.1 h; the drug concentration in the lungs was approximately twice that in the plasma, and the Cmax and AUCt in both plasma and lungs were dose-dependent. 2 Compared with the vehicle group, OPC-167832 (0.625–10 mg/kg, oral administration, 4 weeks) significantly reduced lung CFU, and the lung CFU decreased in a dose-dependent manner within the dose range of 0.625–2.5 mg/kg. 3 OPC-167832 (2.5 mg/kg, intragastric administration, combined with DCMB, 12 weeks) showed the optimal therapeutic effect; the lung CFU of mice was below the detection limit after 6 weeks of treatment, and the lung bacteria of all mice were eradicated after 8 weeks of treatment [1]. |
| Synonyms | Quabodepistat, OPC167832, OPC 167832 |
| Molecular Weight | 456.84 |
| Formula | C21H20ClF3N2O4 |
| Cas No. | 1883747-71-4 |
| Smiles | O(C[C@@]1(O)[C@H](O)CN(CC1)C2=C(F)C=C(Cl)C=C2F)C3=C4C(=C(F)C=C3)NC(=O)CC4 |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. | |||||||||||||||||||||||||||||||||||
| Solubility Information | DMSO: 144 mg/mL (315.21 mM), Sonication is recommended. | |||||||||||||||||||||||||||||||||||
Solution Preparation Table | ||||||||||||||||||||||||||||||||||||
DMSO
Note : The dilution table applies only to solid products. For liquid products, please calculate the stock solution based on the stated concentration and/or density. | ||||||||||||||||||||||||||||||||||||
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 µL PEG300
stock solution to 400 µL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 µL Tween 80 and mix well until fully clarified.
3) Add 450 µL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.