Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
Synonyms:
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 500 μg | $297 | 35 days | 35 days | |
| 1 mg | $568 | 35 days | 35 days | |
| 5 mg | $2,420 | 35 days | 35 days | |
| 10 mg | $4,230 | 35 days | 35 days |
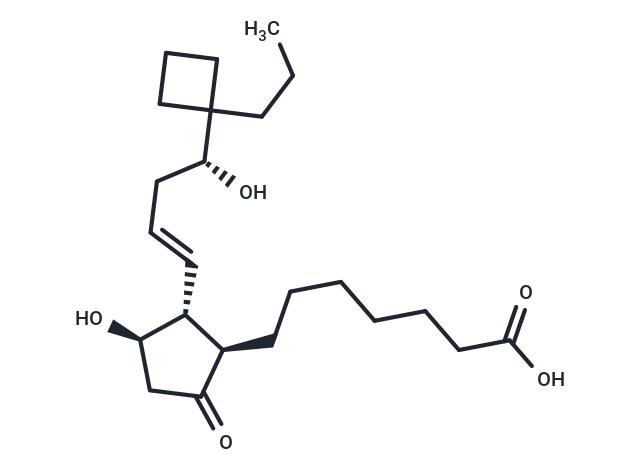
| Description | (R)-Butaprost (free acid) is a prostaglandin E2 (PGE2) analog exhibiting high EP2 receptor subtype selectivity, commonly used to delineate EP receptor expression in human and animal tissues and cells. In 1986, Gardiner induced significant confusion regarding its structure by incorrectly identifying the selective C-16 epimer as (R)-butaprost (refer to the British Journal of Pharmacology, page 46, as TR 4979, and notes). By removing the methyl ester and restoring the native carboxylic acid at C-1, the binding affinity for prostaglandin receptors was enhanced, given such free acids typically display 10 to 100 times greater affinity than their ester counterparts. Although not extensively studied pharmacologically, (R)-butaprost is generally viewed as the less active C-16 epimer, with careful studies conducted later in the United States and Japan ultimately establishing the correct active structure as the 16(S) epimer. |
| Molecular Weight | 394.54 |
| Formula | C23H38O5 |
| Cas No. | 215168-33-5 |
| Smiles | [C@H](C/C=C/[C@@H]1[C@@H](CCCCCCC(O)=O)C(=O)C[C@H]1O)(O)C2(CCC)CCC2 |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. |
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 μL PEG300
stock solution to 400 μL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 μL Tween 80 and mix well until fully clarified.
3) Add 450 μL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.