Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
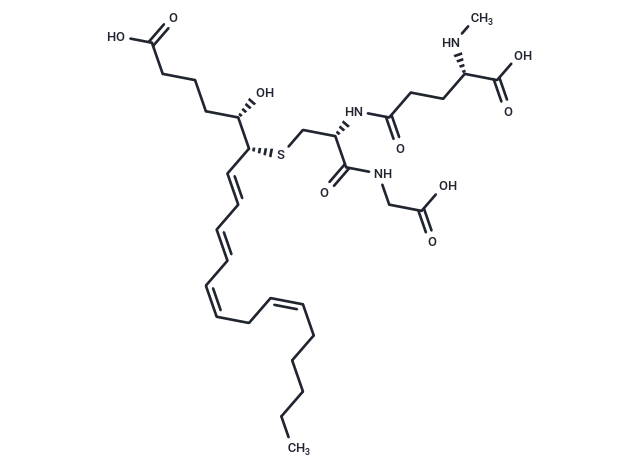
Synonyms: N-methyl Leukotriene C4
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 25 μg | $598 | 35 days | 35 days | |
| 50 μg | $1,130 | 35 days | 35 days | |
| 100 μg | $2,130 | 35 days | 35 days |
| Description | Produced by neutrophils, macrophages, mast cells, and by transcellular metabolism in platelets, leukotriene C4 (LTC4) is the parent cysteinyl leukotriene formed by the LTC4 synthase-catalyzed conjugation of glutathione to LTA4. It is one of the constituents of slow-reacting substance of anaphylaxis (SRS-A) and exhibits potent smooth muscle contracting activity. LTC4, however, is rapidly metabolized to LTD4 and LTE4, which makes the characterization of LTC4 pharmacology difficult. N-methyl Leukotriene C4 (N-methyl LTC4) is a synthetic analog of LTC4 that is not readily metabolized to LTD4 and LTE4.It acts as a potent and selective CysLT2 receptor agonist exhibiting EC50 values of 122 and > 2,000 nM at the human CysLT2 and CysLT1 receptors, respectively. It has essentially the same potency as LTC4 at both the human and murine receptors CysLT2 receptors. N-methyl LTC4 is potent and active in vivo, causing vascular leak in mice overexpressing the human CysLT2 receptor but not in CysLT2 receptor knockout mice. |
| Synonyms | N-methyl Leukotriene C4 |
| Molecular Weight | 639.8 |
| Formula | C31H49N3O9S |
| Cas No. | 131391-65-6 |
| Smiles | [C@@H](CS[C@@H]([C@H](CCCC(O)=O)O)/C=C/C=C/C=C\C/C=C\CCCCC)(NC(CC[C@@H](C(O)=O)NC)=O)C(NCC(O)=O)=O |
| Relative Density. | 1.202 g/cm3 (Predicted) |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. |
| Solubility Information | DMF: 50 mg/mL (78.15 mM), Sonication is recommended. DMSO: 50 mg/mL (78.15 mM), Sonication is recommended. PBS (pH 7.2): 100 μg/mL, Sonication is recommended. Ethanol:H2O (95:5): 2 mg/mL (3.13 mM), Sonication is recommended. Ethanol: 1 mg/mL (1.56 mM), Sonication is recommended. |
Solution Preparation Table | |
Note : The dilution table applies only to solid products. For liquid products, please calculate the stock solution based on the stated concentration and/or density. | |
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 μL PEG300
stock solution to 400 μL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 μL Tween 80 and mix well until fully clarified.
3) Add 450 μL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.