Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
Synonyms:
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 25 mg | Inquiry | 3-6 months | 3-6 months | |
| 50 mg | Inquiry | 3-6 months | 3-6 months | |
| 100 mg | Inquiry | 3-6 months | 3-6 months |
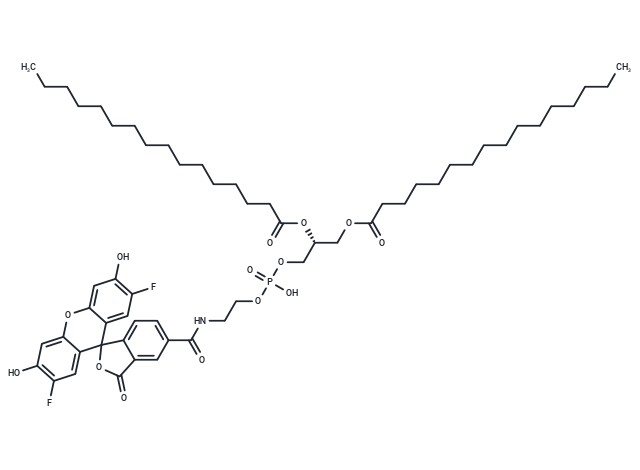
| Description | FG 488 DHPE, a lipid-coupled fluorochrome, is used as the fluorophore Oregon Green 488. It monitors the acidification of lipid vesicles with λex/λem=508/534 nm and is also utilized for quantifying Hv1-induced proton translocation with the same excitation/emission wavelengths [1] [2]. |
| In vitro | FG 488 DHPE exhibits pH-dependent fluorescence emission characteristics [1]. Monitoring acidification in Bulk vesicle assays [1]: 1. Instrument: Jasco FP6500 spectrofluorometer, 37 ℃; excitation at λex=508 nm and emission detection at λem=534 nm. 2. Add 100 μL proteoliposomes (cphospholipid ~60 μM) to 680 μL ATPase buffer with K+-ionophore valinomycin (5 nM) for charge equilibration. 3. Add ATP (1.2 mM) to induce proton pumping. 4. Add 1 mM NaN3 to halt ATP hydrolysis. 5. Add CCCP (carbonyl cyanide 3-chlorophenyl hydrazine, 0.4 μM) to deplete the proton gradient. Conversion into pH-values normalizes fluorescence intensities to those obtained directly after ATP addition. FG 488 DHPE quantifies pH changes induced by the voltage-dependent proton channel Hv1 [2]. Quantification of phospholipid concentrations [2]: 1. Add Perchloric acid (70%, 200 μL) to a sample of unilamellar vesicles containing OG488-DHPE (30 μL). 2. Heat at 220 °C for 60 min to generate inorganic phosphate. 3. Cool to room temperature and add 700 μL of NH4MoO4 (0.45% (w/v)), perchloric acid (12.6% (w/v)), and 700 μL acetic acid (1.7% (w/v)). 4. Obtain a calibration curve for NaH2PO4 concentrations. 5. Incubate samples at 80 °C for 10 min and measure absorption at 820 nm. 6. Calculate phospholipid concentrations using the calibration curve. Proton translocation assay [2]: 1. Instrument: Jasco FP6500 spectrofluorometer, 37 ℃; excitation at λex=508 nm (3 nm band width) and emission detection at λem=534 nm (3 nm band width). 2. Dilute proteoliposomes (POPC/POPG/Chol/OG488-DHPE in 54.5:25:20:0.5 ratio) in buffer A within flux buffer, creating a 14-fold K+-gradient across the membrane. 3. Add valinomycin (13 nM) to induce protonation of OG488-DHPE and quench its fluorescence intensity for active Hv1 channels. 4. Add CCCP (6 nM) to permeabilize vesicles for protons. 5. Plot normalized fluorescence intensity Fnorm as a function of time. Use protein-free vesicles as a control for proton leakage. For experiments with the potential inhibitor 2GBI, dissolve the inhibitor (15 mM) in flux buffer and add (0.5-8.0 μL) to proteoliposomes before valinomycin addition to induce proton translocation. The above information is based on published literature. Experimental procedures should be appropriately modified to meet specific research demands. |
| Molecular Weight | 1086.24 |
| Formula | C58H82F2NO14P |
| Cas No. | 438476-80-3 |
| Smiles | O=C1OC2(C=3C1=CC(C(NCCOP(OC[C@H](OC(CCCCCCCCCCCCCCC)=O)COC(CCCCCCCCCCCCCCC)=O)(=O)O)=O)=CC3)C=4C(OC=5C2=CC(F)=C(O)C5)=CC(O)=C(F)C4 |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. |
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 µL PEG300
stock solution to 400 µL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 µL Tween 80 and mix well until fully clarified.
3) Add 450 µL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.