Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
Synonyms:
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 10 mg | Inquiry | 10-14 weeks | 10-14 weeks | |
| 50 mg | Inquiry | 10-14 weeks | 10-14 weeks |
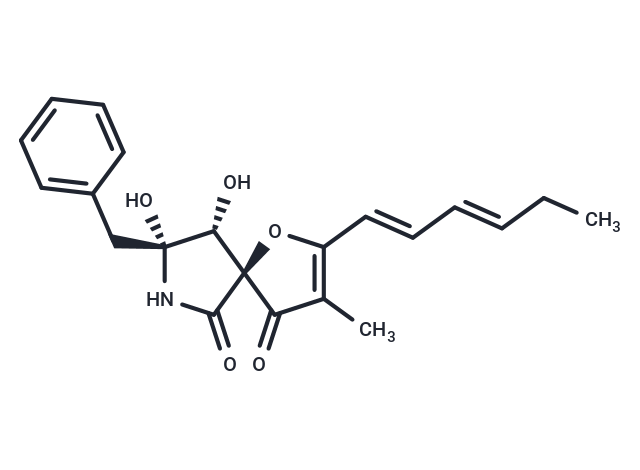
| Description | Azaspirene, a novel angiogenesis inhibitor, is based on the 1-oxa-7-azaspiro[4.4]non-2-ene-4,6-dione framework. Previous studies have shown that Azaspirene effectively inhibits VEGF-induced migration of human umbilical vein endothelial cells (HUVEC), demonstrating a potentially effective dosage. Moreover, azaspirene analogs may exhibit potential antitumor effects against uterine adenocarcinoma, possibly through significant anti-angiogenic activity. |
| Molecular Weight | 369.41 |
| Formula | C21H23NO5 |
| Cas No. | 461644-34-8 |
| Smiles | O[C@@H]1[C@@]2(OC(/C=C/C=C/CC)=C(C)C2=O)C(=O)N[C@]1(CC3=CC=CC=C3)O |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. |
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 μL PEG300
stock solution to 400 μL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 μL Tween 80 and mix well until fully clarified.
3) Add 450 μL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.