 Your shopping cart is currently empty
Your shopping cart is currently empty

Synonyms:
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 5 mg | Inquiry | 8-10 weeks | 8-10 weeks | |
| 50 mg | Inquiry | 8-10 weeks | 8-10 weeks |
| Description | Ferric nitrilotriacetate (Fe-NTA) is a highly active compound formed by the complexation of iron and nitrilotriacetic acid. It is primarily used to induce degenerative diseases through oxidative stress (OS) and can be applied to establish disease models such as acute kidney injury, renal cell carcinoma, liver cancer, and diabetes. |
| In vivo | Ferric nitrilotriacetate can be employed in animal modeling to construct renal carcinoma models. |
| Disease Modeling Protocol | Renal cell carcinoma model
*Precautions: After the last administration of medication to the animal 48He was executed when he was young. |
| Molecular Weight | 243.96 |
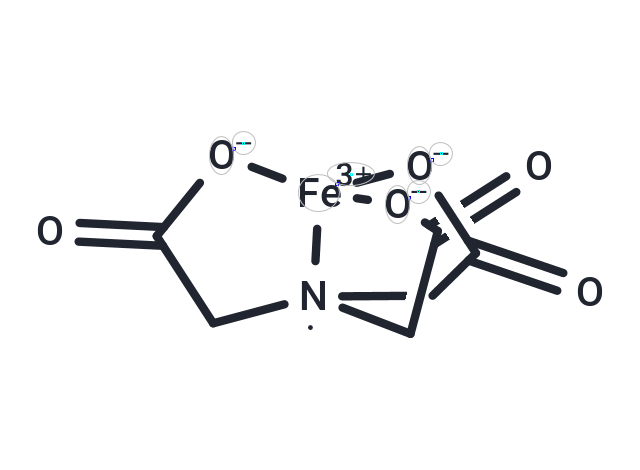
| Formula | C6H6FeNO6 |
| Cas No. | 16448-54-7 |
| Smiles | O=C1[O-][Fe+3]23[N](CC(=O)[O-]2)(CC(=O)[O-]3)C1 |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. |
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 μL PEG300
stock solution to 400 μL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 μL Tween 80 and mix well until fully clarified.
3) Add 450 μL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.