Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
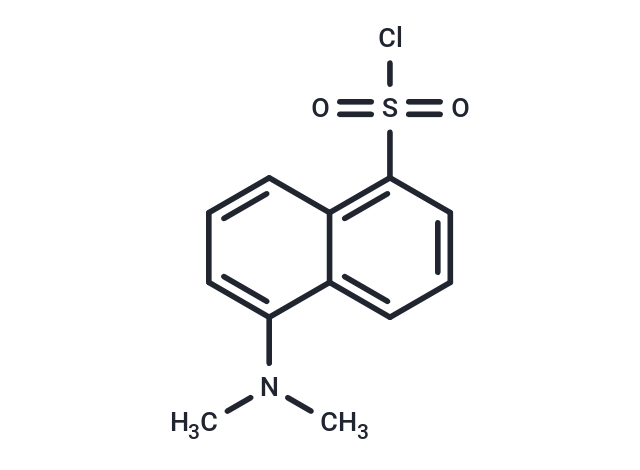
Synonyms: DNSCl
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 1 g | $32 | In Stock | In Stock | |
| 5 g | $91 | - | In Stock |
| Description | Dansyl chloride (DNSC-l) reacts with primary amino groups in both aliphatic and aromatic amines to produce stable blue- or blue-green-fluorescent sulfonamide adducts. It is widely used to modify amino acids, specifically, protein sequencing and amino acid analysis. |
| In vitro | I. Amino acid and protein modification 1. Material preparation (1) DNSC-1 solution: Dissolve DNSC-1 in an appropriate amount of organic solvent (such as acetone or acetonitrile), usually at a concentration of 1-10 mM. (2) Amino acid or protein solution: Prepare the amino acid or protein sample to be modified. The buffer is usually sodium bicarbonate buffer (pH 9-10). (3) Organic solvent: Acetone or acetonitrile is used as the reaction medium. 2. Experimental steps (1) Add DNSC-1 and amino acid/protein solution to the reaction system, ensuring that the molar ratio is 1:1 to 5:1. (2) Incubate at 30-37°C for 30 minutes to 2 hours under light-proof conditions. (3) After the reaction is completed, dilute or separate with an acetonitrile/water mixture. (4) Analyze the product using high performance liquid chromatography (HPLC) or fluorescence spectrometer. II. Amino Acid Analysis 1. Experimental Steps (1) Prepare the amino acid mixture to be analyzed. (2) Add DNSC-1 solution and adjust the pH to alkaline (9-10). (3) After incubation, dilute the product with acetonitrile and directly perform HPLC analysis. (4) On the fluorescence detector, the excitation wavelength is usually 330-350 nm and the emission wavelength is 520-540 nm. 2. Protein N-terminal Sequencing DNSC-1 can react with the N-terminal amino group of the protein to generate fluorescently labeled derivatives for protein sequencing by Edman degradation. Notes: (1) DNSC-1 is sensitive to water and should be kept dry when preparing the solution. (2) The reaction conditions should be kept alkaline (pH 9-10) to ensure efficient derivatization reaction. (3) Operate in a dark environment to avoid degradation of the fluorescent signal. The above information is based on published literature. Experimental procedures should be appropriately modified to meet specific research demands. |
| Synonyms | DNSCl |
| Molecular Weight | 269.75 |
| Formula | C12H12ClNO2S |
| Cas No. | 605-65-2 |
| Smiles | CN(C)c1cccc2c(cccc12)S(Cl)(=O)=O |
| Relative Density. | 1.361 g/cm3 |
| Storage | Keep away from direct sunlight,Keep away from moisture Powder: -20°C for 3 years Shipping with blue ice/Shipping at ambient temperature. | |||||||||||||||||||||||||
| Solubility Information | DMSO: 7.8 mg/mL (28.92 mM), Sonication is recommended. | |||||||||||||||||||||||||
Solution Preparation Table | ||||||||||||||||||||||||||
DMSO
Note : The dilution table applies only to solid products. For liquid products, please calculate the stock solution based on the stated concentration and/or density. | ||||||||||||||||||||||||||
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 µL PEG300
stock solution to 400 µL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 µL Tween 80 and mix well until fully clarified.
3) Add 450 µL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.