Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
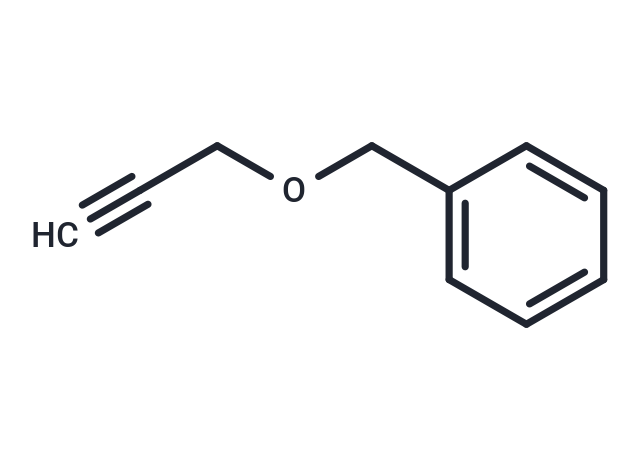
Synonyms: ((Prop-2-yn-1-yloxy)methyl)benzene

| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 5 mg | $50 | - | In Stock | |
| 10 mg | $70 | - | In Stock | |
| 25 mg | $113 | - | In Stock | |
| 50 mg | $163 | - | In Stock | |
| 100 mg | $239 | - | In Stock |
| Description | Benzyl propargyl ether is a versatile synthetic building block characterized by the linear formula C₆H₅CH₂OCH₂C≡CH and defined by its terminal alkyne functionality, which enables participation in diverse carbon–carbon and carbon–heteroatom bond-forming reactions. Benzyl propargyl ether is widely used in the synthesis of substituted carbocyclic aromatic frameworks, pharmaceutical intermediates, and advanced polymeric materials, Benzyl propargyl ether also serves as a valuable precursor in copper(I)-catalyzed azide–alkyne cycloaddition (CuAAC) click chemistry for rapid assembly of functional dendrimers and macromolecules. in carbohydrate chemistry,Benzyl propargyl ether functions as a sterically minimal O-protecting group, and its use at the O-2 position of 4,6-O-benzylidene-protected mannosyl donors enables exceptionally high β-selectivity during challenging glycosylation reactions, supporting efficient synthesis of complex oligosaccharides and glycans. |
| Synonyms | ((Prop-2-yn-1-yloxy)methyl)benzene |
| Molecular Weight | 146.19 |
| Formula | C10H10O |
| Cas No. | 4039-82-1 |
| Smiles | C#CCOCC=1C=CC=CC1 |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. |
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 µL PEG300
stock solution to 400 µL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 µL Tween 80 and mix well until fully clarified.
3) Add 450 µL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.