Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
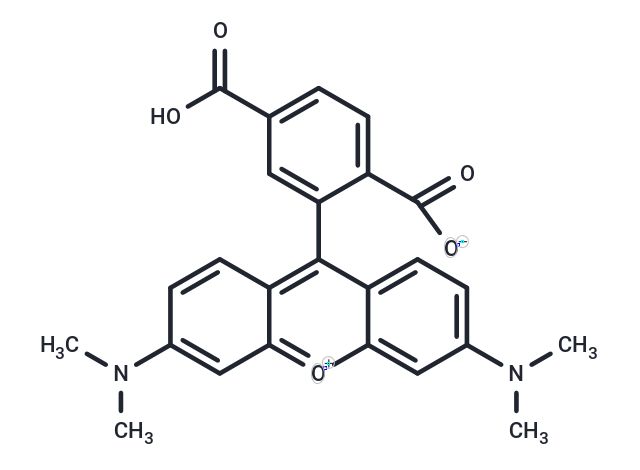
Synonyms: 6-Carboxytetramethylrhodamine
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 10 mg | $41 | - | In Stock | |
| 25 mg | $74 | - | In Stock | |
| 50 mg | $116 | Inquiry | Inquiry |
| Description | 6-TAMRA (6-Carboxytetramethylrhodamine) serves as a popular fluorophore for the creation of bioconjugates, particularly in the synthesis of fluorescent antibodies and avidin derivatives employed in immunochemistry. |
| In vitro | 6-TAMRA labeled nucleotide experiment Operation steps 1. Sample treatment: Incubate in NHOH/methylamine (1:1) solution at 65°C for 10 minutes to separate oligonucleotides from CPG. 2. Take the supernatant and wash CPG with 1 mL EtOH/MeCN/H2O (3:1:1). 3. Mix the supernatants and dry them. 4. Treat with fresh anhydrous triethylammonium fluoride/n-methylpyridone (250 μL, n-methylpyridone 1.5 mL, triethylamine 750 μL, tea-3hf 1.0 mL) at 65°C for 1.5 h to remove t-butyl-dimethylsilyl protecting groups from RNA residues, and add 25 μL 3 M NaOAc and 1 mL n-BuOH to precipitate oligonucleotides. 5. The sample was cooled at -70°C for 1 h and then centrifuged at 10,000 g for 30 min. The supernatant was decanted, washed with aqueous EtOH (70% v/v), and then dried. 6. 6-TAMRA (0.1 mL, 10 mg/mL in dimethyl sulfoxide) was added to the 3'-amino-modified oligonucleotide suspended in 1.0 mL of sodium bicarbonate buffer (pH 8.5) and incubated at 37°C for 12 h. 7. The labeled oligonucleotide was resuspended in water and passed through a G25 Nap-10 disposable desalting column to remove free dye. 8. The oligonucleotide was purified by HPLC with a linear gradient of acetonitrile in 0.1 M triethylammonium acetate (TEAA) buffer, pH 7.2. 9. The entire sample was loaded on a Hamilton PRP-1 column and eluted with a linear gradient of acetonitrile for 40 min. Samples were monitored at 260 and 297 nm, and peaks corresponding to the dual-labeled oligonucleotide species were collected. The above information is based on published literature. Experimental procedures should be appropriately modified to meet specific research demands. |
| Synonyms | 6-Carboxytetramethylrhodamine |
| Molecular Weight | 430.45 |
| Formula | C25H22N2O5 |
| Cas No. | 91809-67-5 |
| Smiles | CN(C)c1ccc2c(-c3cc(ccc3C([O-])=O)C(O)=O)c3ccc(cc3[o+]c2c1)N(C)C |
| Relative Density. | no data available |
| Storage | Keep away from direct sunlight Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. | |||||||||||||||
| Solubility Information | DMSO: 2.86 mg/mL (6.64 mM), Sonication is recommended. | |||||||||||||||
Solution Preparation Table | ||||||||||||||||
DMSO
Note : The dilution table applies only to solid products. For liquid products, please calculate the stock solution based on the stated concentration and/or density. | ||||||||||||||||
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 µL PEG300
stock solution to 400 µL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 µL Tween 80 and mix well until fully clarified.
3) Add 450 µL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.