Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
Synonyms:
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 1 mL | $613 | 4-6 weeks | 4-6 weeks |
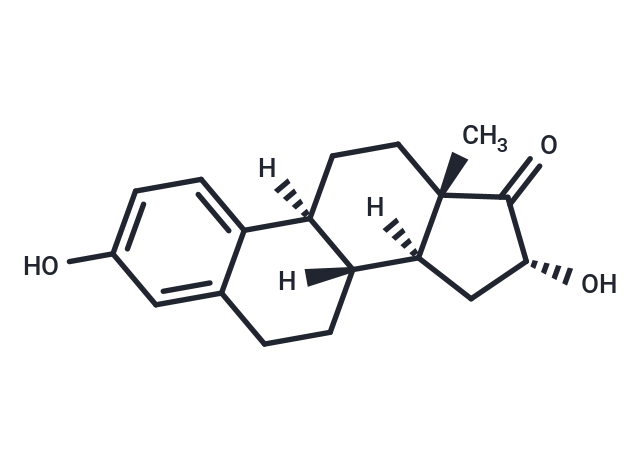
| Description | 16α-Hydroxyestrone (Standard) is a reference standard for research and analysis in studies involving 16α-Hydroxyestrone. The naturally-occurring estrogens are estrone , estradiol , and estriol . 16α-hydroxy Estrone (16α-OHE1) is a hydroxylated metabolite of E1 as well as an interconversion product with E2. E1 is 16α-hydroxylated by cytochrome P450 (CYP) isoforms, including CYP1A1, CYP3A5, CYP3A4, and CYP3A7, with CYP3A5 being breast-specific. 16α-OHE1 is sulphatized or glucuronidated before excretion. It is increased in rheumatoid arthritis and decreased by physical activity. Unlike the parent estrogens and other hydroxylated metabolites of E1, 16α-OHE1 binds covalently and persistently activates estrogen receptors. In addition, this metabolite increases cell proliferation and does not suppress TNF-α secretion, whereas other estrogen metabolites are not pro-proliferative and have marked effects on TNF-α secretion. The levels of 16α-OHE1 are increased in some forms of hormone therapy. Because hormone therapy increases breast cancer risk, 16α-OHE1 has been implicated as a risk factor for breast cancer, although supportive data remains elusive. |
| Molecular Weight | 286.37 |
| Formula | C18H22O3 |
| Cas No. | 566-76-7 |
| Smiles | C[C@@]12[C@]([C@]3([C@@](C=4C(CC3)=CC(O)=CC4)(CC1)[H])[H])(C[C@@H](O)C2=O)[H] |
| Relative Density. | 1.249 g/cm3 (Predicted) |
| Storage | Keep away from direct sunlight Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. |
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 μL PEG300
stock solution to 400 μL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 μL Tween 80 and mix well until fully clarified.
3) Add 450 μL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.