Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
Synonyms: LAP-2, LAP2, HSPN, HSPCAL4, HSPCAL1, HSPCA, HSPC1, HSP90N, HSP90A, Hsp90 α, Hsp90, HSP89A, Hsp89, HSP86, heat shock protein 90kDa α (cytosolic), class A member 1, heat shock protein 90kDa alpha (cytosolic), class A member 1, EL52

| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 5 µg | $48 | - | In Stock | |
| 10 µg | $75 | - | In Stock | |
| 20 µg | $118 | - | In Stock | |
| 50 µg | $229 | 7-10 days | 7-10 days | |
| 100 µg | $386 | - | In Stock | |
| 200 µg | $682 | 7-10 days | 7-10 days | |
| 500 µg | $1,430 | 7-10 days | 7-10 days |
| Bioactivity | Activity testing is in progress. It is theoretically active, but we cannot guarantee it. If you require protein activity, we recommend choosing the eukaryotic expression version first. |
| Description | HSP90 alpha Protein, Human, Recombinant is expressed in E. coli expression system. The predicted molecular weight is 22.6 kDa and the accession number is P07900-1. |
| Species | Human |
| Expression System | E. coli |
| Tag | Tag Free |
| Accession Number | P07900-1 |
| Construction | A DNA sequence encoding the human HSP90 isoform 2 (NP_005339.3) C-terminal segment, corresponding to amino acid sequence (Glu 535-Asp 732) was expressed and purified, with two additonal aa (Gly & Pro) at the N terminus. Predicted N terminal: Gly |
| Protein Purity | ≥ 90 % as determined by SDS-PAGE. ≥ 90 % as determined by SEC-HPLC. 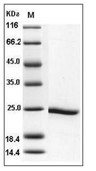 , ,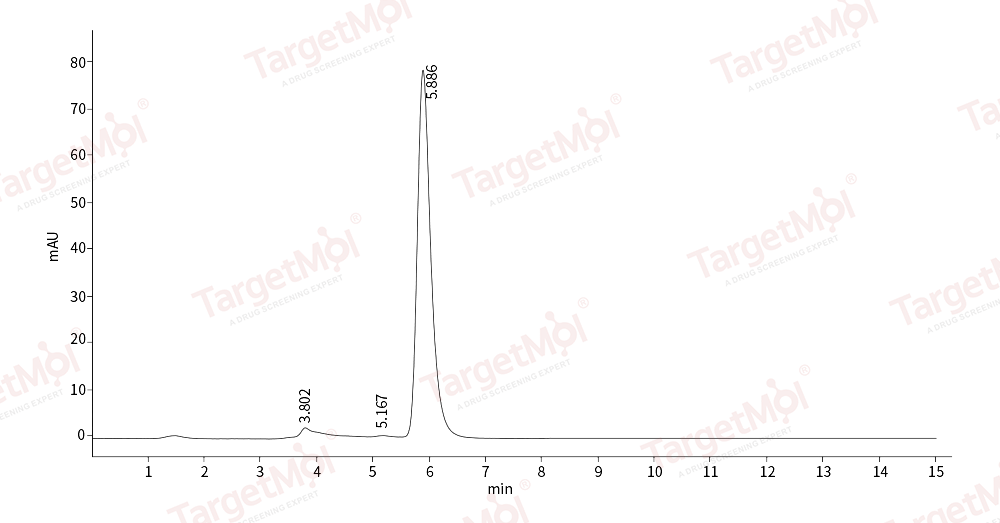 |
| Endotoxin | Please contact us for more information. |
| Formulation | Lyophilized from a solution filtered through a 0.22 μm filter, containing PBS, pH 7.4. Typically, a mixture containing 5% to 8% trehalose, mannitol, and 0.01% Tween 80 is incorporated as a protective agent before lyophilization. |
| Reconstitution | Reconstituted with sterile deionized water to 0.25 mg/mL. Reconstitution conditions may vary depending on the lot. |
| Synonyms | LAP-2, LAP2, HSPN, HSPCAL4, HSPCAL1, HSPCA, HSPC1, HSP90N, HSP90A, Hsp90 α, Hsp90, HSP89A, Hsp89, HSP86, heat shock protein 90kDa α (cytosolic), class A member 1, heat shock protein 90kDa alpha (cytosolic), class A member 1, EL52 |
| Research Background | Heat shock protein 90 (90 kDa heat-shock protein, HSP90) is a molecular chaperone involved in the trafficking of proteins in the cell. It is a remarkably versatile protein involved in the stress response and normal homoeostatic control mechanisms. HSP90 interacts with 'client proteins', including protein kinases, transcription factors, and others, and either facilitates their stabilization and activation or directs them for proteasomal degradation. By this means, HSP90 displays a multifaceted ability to influence signal transduction, chromatin remodeling and epigenetic regulation, development, and morphological evolution. HSP90 operates as a dimer in a conformational cycle driven by ATP binding and hydrolysis at the N-terminus. Disruption of HSP90 leads to client protein degradation and often cell death. Under stressful conditions, HSP90 stabilizes its client proteins and protects the cell against cellular stressors such as in cancer cells. Especially, several oncoproteins act as HSP90 client proteins and tumor cells require higher HSP90 activity than normal cells to maintain their malignancy. For this reason, Hsp90 has emerged as a promising target for anti-cancer drug development. |
| Molecular Weight | 22.6 kDa (predicted); 24 kDa (reducing conditions) |
| Shipping | In general, lyophilized powders are shipped with blue ice, while solutions are shipped with dry ice. |
| Storage | It is recommended to store recombinant proteins at -20°C to -80°C for future use. Lyophilized powders can be stably stored for over 12 months, while liquid products can be stored for 6-12 months at -80°C. For reconstituted protein solutions, the solution can be stored at -20°C to -80°C for at least 3 months. Please avoid multiple freeze-thaw cycles and store products in aliquots. |
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.