Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
Synonyms:
| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 1 mg | $142 | 35 days | 35 days | |
| 5 mg | $663 | 35 days | 35 days | |
| 10 mg | $1,130 | 35 days | 35 days |
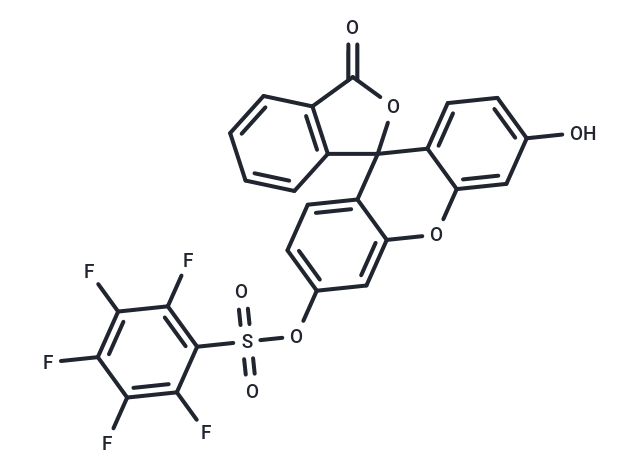
| Description | Pentafluorobenzenesulfonyl fluorescein itself no fluorescence, but were in the H2O2 in sulfonyl key fluoresce when hydrolyzed. Pentafluorobenzenesulfonyl fluorescein is often treated as selective H2O2 sensors, can be used for the detection of H2O2 content in the cell. |
| Cell Research | Instructions 1. Prepare probe solution: PFSF is usually dissolved in anhydrous DMSO to prepare a stock solution (e.g. 1–10 mM), and then diluted to a working concentration (usually 1–10 µM) according to experimental requirements. 2. Sample treatment: 1) For cell experiments, PFSF is added to the culture medium (usually used in phenol red-free culture medium containing 1% FBS to avoid fluorescence background interference), and then incubate the cells for 30 minutes to 1 hour at 37°C, away from light. 2) For chemical or solution experiments, PFSF can be directly mixed with the target system (such as a buffer solution containing H₂O₂). 3. Fluorescence detection: Fluorescence excitation wavelength: 490–500 nm, emission wavelength: 515–530 nm. The signal can be recorded using a fluorescence microscope, fluorescence spectrophotometer, or fluorescence plate reader. 4. Calibration and control: 1) It is recommended to set up a no-H₂O₂ control group and a H₂O₂ standard curve to verify signal specificity. 2) To ensure probe specificity, catalase can be added to eliminate H₂O₂ interference. Notes 1) Storage conditions: PFSF should be stored at -20°C in the dark and fully dissolved before use. 2) Experimental environment: Avoid high temperature and direct exposure to strong light to prevent probe decomposition or increased autofluorescence. 3) Selectivity verification: In addition to H₂O₂, the probe may have a slight reaction to other strong oxidants (such as peroxynitrite), and the specificity should be verified according to experimental requirements. The above information is based on published literature. Experimental procedures should be appropriately modified to meet specific research demands. |
| Molecular Weight | 562.42 |
| Formula | C26H11F5O7S |
| Cas No. | 728912-45-6 |
| Smiles | O=C1OC2(C3=CC=C(O)C=C3OC4=CC(OS(=O)(=O)C=5C(F)=C(F)C(F)=C(F)C5F)=CC=C42)C=6C=CC=CC16 |
| Relative Density. | no data available |
| Storage | Store at low temperature,Keep away from direct sunlight Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. |
| Solubility Information | DMSO:PBS (pH 7.2)(1:1): 0.25 mg/mL (0.44 mM), Sonication is recommended. DMSO: 30 mg/mL (53.34 mM), Sonication is recommended. Ethanol: 20 mg/mL (35.56 mM), Sonication is recommended. DMF: 30 mg/mL (53.34 mM), Sonication is recommended. |
Dissolve 2 mg of the compound in 100 μL DMSO![]() to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
to obtain a stock solution at a concentration of 20 mg/mL . If the required concentration exceeds the compound's known solubility, please contact us for technical support before proceeding.
1) Add 100 μL of the DMSO![]() stock solution to 400 μL PEG300
stock solution to 400 μL PEG300![]() and mix thoroughly until the solution becomes clear.
and mix thoroughly until the solution becomes clear.
2) Add 50 μL Tween 80 and mix well until fully clarified.
3) Add 450 μL Saline,PBS or ddH2O![]() and mix thoroughly until a homogeneous solution is obtained.
and mix thoroughly until a homogeneous solution is obtained.
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.