- Remove All
 Your shopping cart is currently empty
Your shopping cart is currently empty
(±)5-iPF2α-VI
Isoprostanes are prostaglandin (PG)-like products of free-radical induced lipid peroxidation. Although the isoprostanes derived from arachidonic acid are the best characterized, many other polyunsaturated fatty acids can form isoprostanes. (±)5-iPF2α-VI is one of dozens of possible stereo- and regioisomeric isoprostanes which can be formed from arachidonic acid. To date, the most extensively studied of these is 8-isoprostane (8-epi-PGF2α, iPF2α-III). However, 8-isoprostane is a minor isoprostane constituent when compared to some of the other isomers which form in natural conditions of oxidative stress. (±)5-iPF2α-VI is an isoprostane from the unique Type VI class of isoprostanes. This class has been shown to be one of the major isoprostane products, in contrast to 8-isoprostane. In addition to being produced in greater abundance than 8-isoprostane, Type VI isoprostanes form internal lactones, which facilitates their extraction and purification from biological samples.
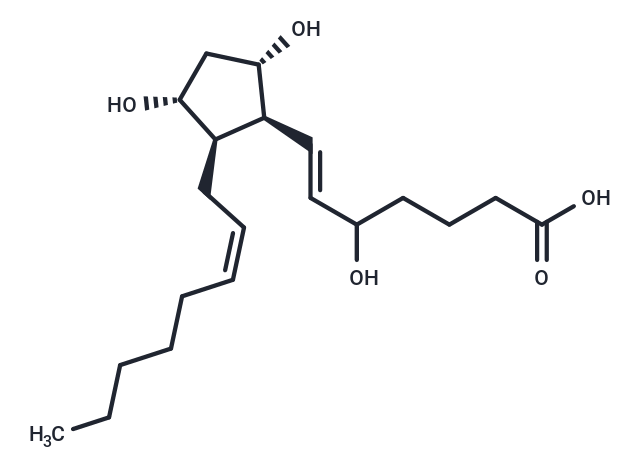
(±)5-iPF2α-VI
| Pack Size | Price | Availability | Quantity |
|---|---|---|---|
| 10 μg | TBD | 35 days | |
| 25 μg | TBD | 35 days | |
| 50 μg | TBD | 35 days | |
| 100 μg | TBD | 35 days |
Product Introduction
| Description | Isoprostanes are prostaglandin (PG)-like products of free-radical induced lipid peroxidation. Although the isoprostanes derived from arachidonic acid are the best characterized, many other polyunsaturated fatty acids can form isoprostanes. (±)5-iPF2α-VI is one of dozens of possible stereo- and regioisomeric isoprostanes which can be formed from arachidonic acid. To date, the most extensively studied of these is 8-isoprostane (8-epi-PGF2α, iPF2α-III). However, 8-isoprostane is a minor isoprostane constituent when compared to some of the other isomers which form in natural conditions of oxidative stress. (±)5-iPF2α-VI is an isoprostane from the unique Type VI class of isoprostanes. This class has been shown to be one of the major isoprostane products, in contrast to 8-isoprostane. In addition to being produced in greater abundance than 8-isoprostane, Type VI isoprostanes form internal lactones, which facilitates their extraction and purification from biological samples. |
| Alias | (±)5-iPF2α-VI |
| Molecular Weight | 354.487 |
| Formula | C20H34O5 |
| Cas No. | 179094-11-2 |
| Relative Density. | 1.153 g/cm3 (Predicted) |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year | Shipping with blue ice. | ||||||||||||||||||||||||||||||||||||||||
| Solubility Information | DMF: 50 mg/mL (141.05 mM), Sonication is recommended. DMSO: 50 mg/mL (141.05 mM), Sonication is recommended. Ethanol: 50 mg/mL (141.05 mM), Sonication is recommended. PBS (pH 7.2): 0.5 mg/mL (1.41 mM), Sonication is recommended. | ||||||||||||||||||||||||||||||||||||||||
Solution Preparation Table | |||||||||||||||||||||||||||||||||||||||||
PBS (pH 7.2)/DMF/DMSO/Ethanol
DMF/DMSO/Ethanol
| |||||||||||||||||||||||||||||||||||||||||
Sci Citations
Calculator
In Vivo Formulation Calculator (Clear solution)
Dose Conversion
Tech Support

Copyright © 2015-2025 TargetMol Chemicals Inc. All Rights Reserved.




