Shopping Cart
Remove All Your shopping cart is currently empty
Your shopping cart is currently empty
Synonyms: TBE-1, MONA, MMP-II, MMP-2, matrix metallopeptidase 2, CLG4A, CLG4

| Pack Size | Price | USA Stock | Global Stock | Quantity |
|---|---|---|---|---|
| 5 µg | $182 | - | In Stock | |
| 10 µg | $303 | - | In Stock | |
| 20 µg | $485 | In Stock | In Stock | |
| 50 µg | $970 | - | In Stock | |
| 100 µg | $1,485 | - | In Stock | |
| 200 µg | $2,330 | 7-10 days | 7-10 days | |
| 500 µg | $4,670 | 7-10 days | 7-10 days |
| Bioactivity | 1. Measured by its ability to cleave the fluorogenic peptide substrate Mca-PLGL-Dpa-AR-NH2. The specific activity is > 1,000 pmoles/min/µg. (Activation description: The proenzyme needs to be activated by APMA for an activated form)
2. Measured by its binding ability in a functional ELISA. Immobilized Human MMP-2 at 2 μg/ml (100 μl/well) can bind Human TIMP2 hFc, the EC50 of Human TIMP2 hFc is 6.0-30.0 ng/mL. |
| Description | MMP-2 Protein, Human, Recombinant is expressed in HEK293 mammalian cells. The predicted molecular weight is 72 kDa and the accession number is A0A024R6R4. |
| Species | Human |
| Expression System | HEK293 Cells |
| Tag | Tag Free |
| Accession Number | P08253 |
| Construction | A DNA sequence encoding the native human MMP2 (NP_004521.1) (Met 1-Cys 660) was expressed and purified. Predicted N terminal: Ala 30 |
| Protein Purity | ≥ 90 % as determined by SDS-PAGE ≥ 90 % as determined by SEC-HPLC. 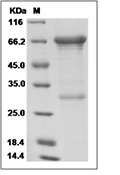 , ,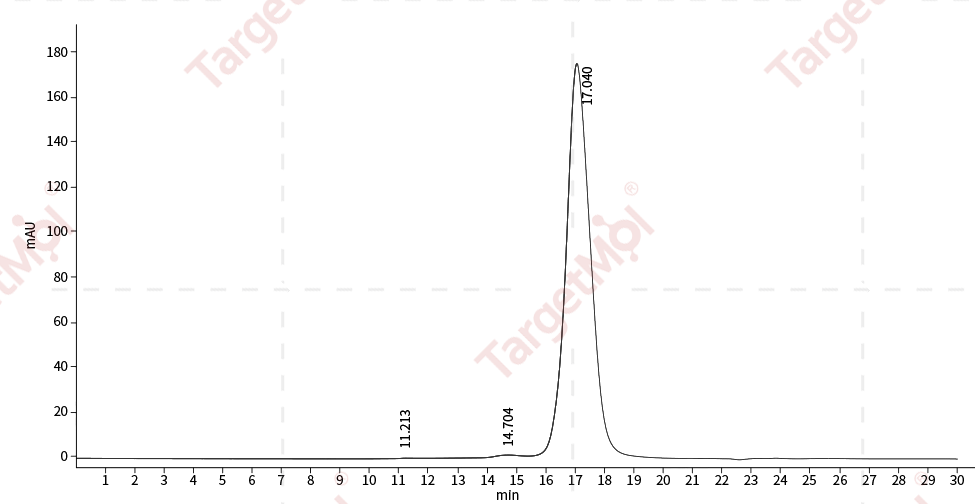 |
| Endotoxin | < 1.0 EU/μg of the protein as determined by the LAL method. |
| Formulation | Lyophilized from a solution filtered through a 0.22 μm filter, containing 0.05% Brij-35, 150 mM NaCl, 5 mM CaCl2, 50 mM Tris, pH 7.5. Typically, a mixture containing 5% to 8% trehalose, mannitol, and 0.01% Tween 80 is incorporated as a protective agent before lyophilization. |
| Reconstitution | Reconstituted with sterile deionized water to 0.25 mg/mL. Reconstitution conditions may vary depending on the lot. |
| Synonyms | TBE-1, MONA, MMP-II, MMP-2, matrix metallopeptidase 2, CLG4A, CLG4 |
| Research Background | Matrix Metalloproteinase-2 (MMP-2) is an enzyme that degrades components of the extracellular matrix and thus plays a pivotal role in cell migration during physiological and pathological processes. MMP-2 expression is dependent on extracellular matrix metalloproteinase inducer (EMMPRIN), Her2/neu, growth factors, cytokines, and hormones. Pro-MMP-2 activation needs MT1-MMP and TIMP-2 contribution. MMP-2 is changed in distribution and increased in amount in the ventral cochlear nucleus after unilateral cochlear ablation. A low level of MMP-2 is linked to a favorable prognosis in patients with a hormone receptor-negative tumor, usually associated with high risk. As a zymogen requiring proteolytic activation for catalytic activity, MMP-2 has been implicated broadly in the invasion and metastasis of many cancer model systems, including human breast cancer (HBC). Blocking MMP-2 secretion and activation during breast carcinoma development may decrease metastasis. The detection of active MMP-2 alone or the rate of pro-MMP-2 and active MMP-2 is considered a very sensitive indicator of cancer metastasis. Modulation of MMP-2 expression and activation through specific inhibitors and activators may thus provide a new mechanism for breast cancer treatment. |
| Molecular Weight | 72 kDa (reducing conditions) |
| Shipping | In general, lyophilized powders are shipped with blue ice, while solutions are shipped with dry ice. |
| Storage | It is recommended to store recombinant proteins at -20°C to -80°C for future use. Lyophilized powders can be stably stored for over 12 months, while liquid products can be stored for 6-12 months at -80°C. For reconstituted protein solutions, the solution can be stored at -20°C to -80°C for at least 3 months. Please avoid multiple freeze-thaw cycles and store products in aliquots. |
| Size | Quantity | Unit Price | Amount | Operation |
|---|

Copyright © 2015-2026 TargetMol Chemicals Inc. All Rights Reserved.